News
TPCB News Archives
October 2024
TPCB Welcomes Steve Bonilla and Elizabeth Campbell to the Faculty
TPCB students have unparalleled opportunities to train with faculty on forefront research across the spectrum of chemical biology. Two new faculty have recently joined the program, bringing expertise in RNA structure and microbial pathogenesis.
 • Prof. Steve Bonilla recently launched his lab at Rockefeller and joined the TPCB faculty. His lab studies the three-dimensional structures of RNA and how they help viruses replicates. In particular, he seeks to understand how viruses use RNA 3D structures to recruit RNA-dependent RNA polymerases to the viral genome, leveraging cryo-electron microscopy. He is also developing methods to visualize RNA molecules that dynamically shift between multiple 3D structures, and to predict their response to small-molecule binding and changes in the environment. This work combines structural and computational approaches and has implications for viral and cellular RNA biology. Prof. Bonilla completed his PhD studies at Stanford University and postdoctoral training as an HHMI Hanna Gray Fellow at the University of Colorado Anschutz Medical Campus. [Read more at: Rockefeller News | TPCB]
• Prof. Steve Bonilla recently launched his lab at Rockefeller and joined the TPCB faculty. His lab studies the three-dimensional structures of RNA and how they help viruses replicates. In particular, he seeks to understand how viruses use RNA 3D structures to recruit RNA-dependent RNA polymerases to the viral genome, leveraging cryo-electron microscopy. He is also developing methods to visualize RNA molecules that dynamically shift between multiple 3D structures, and to predict their response to small-molecule binding and changes in the environment. This work combines structural and computational approaches and has implications for viral and cellular RNA biology. Prof. Bonilla completed his PhD studies at Stanford University and postdoctoral training as an HHMI Hanna Gray Fellow at the University of Colorado Anschutz Medical Campus. [Read more at: Rockefeller News | TPCB]
 • Prof. Elizabeth Campbell recently joined the faculty of TPCB and Rockefeller as the Corinne P. Greenberg Women & Science Professor. Her lab studies the molecular mechanisms of pathogenesis in Mycobacterium tuberculosis and SARS-CoV-2, with a particular focus on the biochemistry and structure of microbial transcription, and how small-molecule drugs can target these processes effectively. Prof. Campbell completed her PhD and postdoctoral studies at Rockefeller and co-led the laboratory of TPCB faculty member Seth Darst before launching her own lab this fall. A native of Guyana, she grew up in Queens and was recently awarded the inaugural Inclusive Excellence Award from Rockefeller’s office of Diversity, Equality, and Inclusion for her advocacy of diversity in science and outstanding mentorship of undergraduate and graduate students. [Read more at: Rockefeller News | TPCB]
• Prof. Elizabeth Campbell recently joined the faculty of TPCB and Rockefeller as the Corinne P. Greenberg Women & Science Professor. Her lab studies the molecular mechanisms of pathogenesis in Mycobacterium tuberculosis and SARS-CoV-2, with a particular focus on the biochemistry and structure of microbial transcription, and how small-molecule drugs can target these processes effectively. Prof. Campbell completed her PhD and postdoctoral studies at Rockefeller and co-led the laboratory of TPCB faculty member Seth Darst before launching her own lab this fall. A native of Guyana, she grew up in Queens and was recently awarded the inaugural Inclusive Excellence Award from Rockefeller’s office of Diversity, Equality, and Inclusion for her advocacy of diversity in science and outstanding mentorship of undergraduate and graduate students. [Read more at: Rockefeller News | TPCB]
September 2024
Christian Baca Recognized by the Max Birnstiel Foundation
 TPCB student Christian Baca has been recognized for his research with an Honourable Mention for the 2024 International Birnstiel Awards for Doctoral Research in Molecular Life Sciences. The awards celebrate exceptional talent in molecular life sciences and reward early achievements of young scientists. They are sponsored by the Max Birnstiel Foundation and the Research Institute of Molecular Pathology (IMP) in Vienna, Austria. Christian’s thesis research with Prof. Luciano Marraffini at Rockefeller focuses on characterizing the molecular systems that bacteria use to defend themselves from viral infection by bacteriophages. These systems, called CRISPR-Cas, recognize foreign nucleic acids, triggering an adaptive defense response in the cell. In most cases, this defense involves degrading the viral DNA or RNA. However, Christian has discovered new mechanisms by which bacteria combat the viruses by causing the infected cell to sacrifice itself and prevent viral propagation. This work has been carried out in collaboration with Dr. Puja Majumder, a postdoctoral fellow in the lab of TPCB Emeritus Faculty Member Prof. Dinshaw Patel, and published in part in the journal Nature. [Read more at: Birnstiel Awards]
TPCB student Christian Baca has been recognized for his research with an Honourable Mention for the 2024 International Birnstiel Awards for Doctoral Research in Molecular Life Sciences. The awards celebrate exceptional talent in molecular life sciences and reward early achievements of young scientists. They are sponsored by the Max Birnstiel Foundation and the Research Institute of Molecular Pathology (IMP) in Vienna, Austria. Christian’s thesis research with Prof. Luciano Marraffini at Rockefeller focuses on characterizing the molecular systems that bacteria use to defend themselves from viral infection by bacteriophages. These systems, called CRISPR-Cas, recognize foreign nucleic acids, triggering an adaptive defense response in the cell. In most cases, this defense involves degrading the viral DNA or RNA. However, Christian has discovered new mechanisms by which bacteria combat the viruses by causing the infected cell to sacrifice itself and prevent viral propagation. This work has been carried out in collaboration with Dr. Puja Majumder, a postdoctoral fellow in the lab of TPCB Emeritus Faculty Member Prof. Dinshaw Patel, and published in part in the journal Nature. [Read more at: Birnstiel Awards]
August 2024
TPCB Hosts 20th Annual Tri-I Chemical Biology Symposium
For two decades, the annual Tri-Institutional Chemical Biology Symposium has showcased the latest advances across the field of chemical biology. This year’s event, which is hosted and organized by TPCB students, was held on August 14, 2024 on the Rockefeller University campus. The symposium included seminars by eminent guest faculty speakers as well as several TPCB students, and a vibrant poster session featuring over 60 contributions from across the community. Over 225 scientists participated in the meeting, and the seminars were webcast for those unable to attend in person.


[1st photo]: Prof. Yamuna Krishnan presents a faculty seminar on calcium transport. [2nd photo]: TPCB student Christopher Nieves Escobar presents a short talk on photoredox catalysis.
The symposium kicked off with a faculty presentation by Prof. Yamuna Krishnan of the University of Chicago on her studies of lysosomal calcium transport, in which her lab has developed a multifunctional DNA-based nanosensor to discover a lysosomal calcium importer, Lci-1. TPCB student Lauren Vostal then spoke about her work with Prof. Tarun Kapoor at Rockefeller on investigations of VCP, an AAA-family protein that unfolds ubiquitinated proteins en route to proteasomal degradation, in which she used amber suppression to install photo crosslinking residues in VCP and identified deubiquitinase binding partners, one of which was characterized in detail using cryo-electron microscopy and biophysical methods. Next, Christopher Nieves Escobar, a TPCB student in the lab of Prof. Derek Tan at MSK, shared his research on 3-oxetanols as bioisosteres for carboxylic acids, in which he has developed a novel photoredox-catalyzed reaction to convert carboxylic acid drugs directly into 3-oxetanol analogues for biological evaluation. The morning session closed out with a faculty talk by Prof. Jonathan Long from Stanford University, who presented his lab’s recent studies of the orphan metabolite N-acetyltaurine, in which they have used metabolomics and human genetics to identify the enzyme that hydrolyzes this metabolite, PTER, as well as an exciting role for this metabolite in regulating body weight.
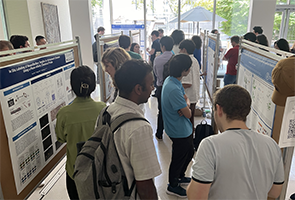

[1st photo]: The poster session featured over 50 presentations across the spectrum of chemical biology. [2nd photo]: TPCB student Marcell Simon presents a poster on his undergraduate research at Pitzer College.
After a lunch break, over 50 students, postdocs, and staff scientists presented posters on their research across the full range of chemical biology. These diverse projects included work on environmental DNA libraries, GPCR interactome mapping, single-molecule analysis of transcription, molecular mechanisms of inflammasome activation, ion channel inhibitors for Parkinson’s disease, computational modeling of SARS-CoV-2 main protease inhibitors, natural product antibiotics, novel drug targets for cystic fibrosis, allosteric inhibitors of RAF-family complexes, nanobody-based protein tags, nanoparticle degraders of nuclear hormones, small-molecule effectors of microbiome–host interactions, and structure-based drug design of AAA proteins, among many others. TPCB faculty members and the visiting keynote speakers served as poster judges and selected awardees in four categories, with poster prizes provided by TPCB and our promotional partners.

[1st photo]: TPCB student Kristen Vogt discusses her work with Prof. David Scheinberg at MSK. [2nd photo]: TPCB faculty members Prof. Viviana Risca and Prof. Daniel Heller view poster presentations.
In afternoon session, Prof. Heeseon An, a TPCB faculty member at MSK, presented her lab’s development of a chemical proteomic approach to measure changes in rates of protein degradation in response to stimuli, which led to the discovery of HMGCS1 as a novel nexus that connects mTOR’s nutrient sensing to sterol metabolism. TPCB student Alexa Strauss then shared her work with Prof. Joshua Levitz at Weill Cornell on the structural basis for biased signaling in a synpatic GPCR, mGluR, by positive allosteric modulators, leveraging cryo-electron microscopy. The final faculty seminar was given by Prof. Carlos Bustamante of UC Berkeley, who discussed his lab’s recent work using optical tweezers to understand co-translational protein folding by following the birth and growth of a protein on the surface of a ribosome in real time.


[1st photo]: Prof. Jonathan Long discusses his recent investigations of N-acetyltaurine in body weight regulation. [2nd photo]: TPCB faculty member Prof. Heeseon An presents her work using comparative degradomics to discover a new component of the nutrient sensing pathway.
This 20th annual symposium was organized and hosted by TPCB students Anoosha Banerjee, Karl Lin, and Kaylyn Spotton, with support from faculty mentor Prof. Ekaterina Vinogradova, TPCB Director Prof. Derek Tan, and TPCB Program Assistant Anna Rejno. Seminars were webcast with support from Rockefeller University. Generous support was provided by promotional partners ACS Chemical Biology, Biochemistry, Cell Chemical Biology, ChemBioChem, Chemical Science, Nature Chemical Biology, Organic & Biomolecular Chemistry, RSC Chemical Biology, and the New York Academy of Sciences.


[1st photo]: Symposium speakers and organizers (from left): TPCB Director Prof. Derek Tan, Christopher Nieves Escobar, Anoosha Banerjee, Alexa Strauss, Lauren Vostal, Kaylyn Spotton, Karl Lin, Prof. Yamuna Krishnan, TPCB Faculty Mentor Prof. Ekaterina Vinogradova, Prof. Heeseon An, and Prof. Jonathan Long. [2nd photo]: Winners of the Student Poster Awards (from left): TPCB Director Prof. Derek Tan, Nina Fox, Yang Xiao, Charles Warren, Giorgos Hiotis, Marcell Simon, Ruijie Xiang, Pooja Pandya, and TPCB Faculty Mentor Prof. Ekaterina Vinogradova.
Poster Award Winners
Early Stage Students
• Marcell Simon, Pitzer College (Aaron Leconte Lab)
– Discovery of Red-Shifting Mutations in Firefly Luciferase Using High-Throughput
Biochemistry
– ChemBioChem Prize
• Ruijie Xiang, The Rockefeller University (Paul Cohen Lab)
– Cargo Identification and Regulatory Mechanisms of Endolysosome Exocytosis in Adipocytes
– RSC Chemical Biology Prize
• Nina Fox, Memorial Sloan Kettering (Omar Abdel-Wahab Lab)
– Therapeutic Modulation of Surface CD47 Abundance via Targeting the Dual-specificity Kinase DYRK1A
– Cell Chemical Biology Prize
TPCB Graduate Students
• Yang Xiao, TPCB (David Lab, MSK)
– Deciphering Histone Glycation in Cancer Using Novel Site-Specific Antibodies
– ChemBioChem Prize
• Charles Warren, TPCB (Geri Lab, Weill Cornell)
– Global Protein-Ligand Affinity Mapping Using Proximity Labeling
– RSC Organic & Biomolecular Chemistry Prize
• Giorgos Hiotis, TPCB (Walz Lab, Rockefeller)
– Different Interactions with the Pocket Lipids Explain the Poor Mechanosensitivity of YnaI Compared to MscS
– Nature Chemical Biology Prize
• Linzhi Ye, TPCB (Brady Lab, Rockefeller)
– Fusobacteria Induce Inflammatory Responses via Simultaneous Release of ADP-Heptose and Ribonucleotides
– Nature Chemical Biology Prize
• Lucas Repeta, TPCB (Lima Lab, MSK)
– MTREC: Activities and Architectures of an RNA Exosome-Associated Helicase Complex
– Cell Chemical Biology Prize
Open Graduate Students
• Ersilia Barin, Weill Cornell Pharmacology (An Lab, MSK)
– Rapid Degradation of Ribosomal Proteins, Ribo-DART, Determines the Role of r-Proteins after Ribosome Biogenesis
– ChemBioChem Prize
• Pooja Pandya, Weill Cornell Pharmacology (Tan Lab, MSK)
– Synthesis of Copper-Chelating Diisonitrile Natural Products from M. tuberculosis
– Chemical Science Prize
Postdoctoral Fellows
• SeCheol Oh, PhD, Memorial Sloan Kettering (Richard Hite Lab)
– Discovery of Selective Inhibitors for the Lysosomal Parkinson’s Disease Channel TMEM175
– RSC Chemical Biology Prize


[1st photo]: Attendees attend the post-symposium reception. [2nd photo]: TPCB faculty member and speaker Prof. Heeseon An and TPCB student Nico Carbone enjoy the symposium dinner with colleagues.
Tri-I Chemical Biology Summer Program Hosts Undergraduates in TPCB Labs
 The Tri-Institutional Chemical Biology Summer Program (ChBSP) has developed into a premier venue for aspiring undergraduate scientists to gain hands-on laboratory research experience at the forefront of chemical biology. The program hosted 6 outstanding summer interns this year from across the country. In addition to working with TPCB students and faculty in the labs, they attended scientific and career development presentations, participated in a journal club hosted by TPCB students, and enjoyed outings in NYC with other summer interns. The program concluded with a poster session on August 8, 2024, where the students shared their research discoveries, presenting along with students from a variety of other summer programs at MSK.
The Tri-Institutional Chemical Biology Summer Program (ChBSP) has developed into a premier venue for aspiring undergraduate scientists to gain hands-on laboratory research experience at the forefront of chemical biology. The program hosted 6 outstanding summer interns this year from across the country. In addition to working with TPCB students and faculty in the labs, they attended scientific and career development presentations, participated in a journal club hosted by TPCB students, and enjoyed outings in NYC with other summer interns. The program concluded with a poster session on August 8, 2024, where the students shared their research discoveries, presenting along with students from a variety of other summer programs at MSK.
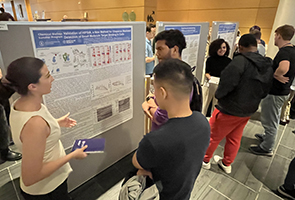
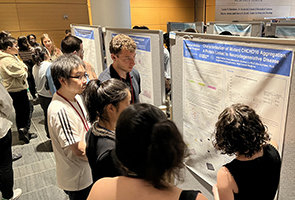
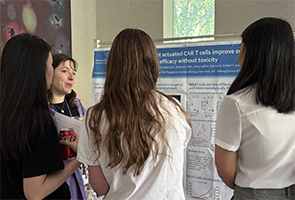
Since its inception in 2018, ChBSP has been led by Prof. Daniel Bachovchin, a TPCB faculty member, and Dr. Ushma Neill, MSK Vice Precident of Scientific Education and Training. The program receives generous financial support from the Sanders Tri-Institutional Discovery Institute, which is directed by Dr. Peter Meinke, with additional support from Memorial Sloan Kettering Cancer Center, the Rockefeller University, and Weill Cornell Graduate School. This year’s journal club was hosted by TPCB student Charles Warren (himself a ChBSP alum), with Michelle Guo, Jared Ramsey, and Alexa Strauss.






Katelyn Dale, University of California, Berkeley, Class of 2024
Synthesis of ASPIR Alk-2 for Developing Selective Chemical Inhibitors of AAA Mechanoenzymes, with Prof. Tarun Kapoor, Rockefeller
Chloe Fields, Boston College, Class of 2024
Phenotypic and Target-based Screening for Inhibition of Macrophage Activation, with Prof. Ekaterina Vinogradova, Rockefeller
Shira Freilich, Vassar College, Class of 2024
Characterization of Mutant CHCHD10 Aggregation: A Protein Linked to Neurodegenerative Disease, with Prof. David Eliezer, Weill Cornell
Angelina Kramer, Penn State University, Class of 2024
Validation of HIPStA, a New Method for Chaperone Mediated Detection of Small Molecule Target Binding in Cells, with Prof. Arvin Dar, MSK
Cristina Marrero Robles, University of Puerto Rico, Rio Piedras, Class of 2025
Exploring and Characterizing PDXK as an Imiquimod Target, with Prof. Daniel Bachovchin, MSK
Giulliano Richetta, Pomona College, Class of 2024
Unveiling the Dark Proteome: Using µMap as a High-throughput Peptide Screening Tool , with Prof. Jacob Geri, Weill Cornell
Group photo provided by Richard Dewitt, MSK Marketing & Communication.
July 2024
Gabriella Chua Recognized with 2024 TPCB Service Award
 TPCB students have played a leading role in building a strong culture of service in our scientific community. These efforts include scientific events, outreach, peer mentoring, and training programs that impact not only to students on our Tri-Institutional campuses, but around the world. The TPCB Student Service Award was established last year to recognize this important work, and Gabriella Chua has been named the 2024 honoree. Gabriella has served in numerous leadership roles within TPCB and the broader scientific community. In 2021, she was a student Co-Organizer of the Tri-Institutional Chemical Biology Symposium, the premier scientific event sponsored by TPCB. She has also served on the TPCB Student Organizing Committee, which oversees our graduate student recruitment efforts, since 2020 and chaired the committee in 2021–2022. Further, Gabriella served as Co-President of the High School Catalyst Program at Weill Cornell, a hands-on research program for aspiring young scientists from 2020–2022. In that vein, she spearheaded successful efforts to secure external funding and matching funds from the graduate school to provide stipends to these students during their participation in the program. Gabriella also launched the “Asian Perspectives on the Hidden Curriculum” discussion series, sponsored by the Rockefeller Office of Diversity, Equity, and Inclusion Microgrant Program, which aims to address training inequities and to build a stronger Asian community in the sciences. Finally, she has also engaged actively in numerous science outreach programs sponsored by the New York Academy of Sciences, including as an Afterschool STEM Teacher, a Scientist in Residence, and a Panelist.
TPCB students have played a leading role in building a strong culture of service in our scientific community. These efforts include scientific events, outreach, peer mentoring, and training programs that impact not only to students on our Tri-Institutional campuses, but around the world. The TPCB Student Service Award was established last year to recognize this important work, and Gabriella Chua has been named the 2024 honoree. Gabriella has served in numerous leadership roles within TPCB and the broader scientific community. In 2021, she was a student Co-Organizer of the Tri-Institutional Chemical Biology Symposium, the premier scientific event sponsored by TPCB. She has also served on the TPCB Student Organizing Committee, which oversees our graduate student recruitment efforts, since 2020 and chaired the committee in 2021–2022. Further, Gabriella served as Co-President of the High School Catalyst Program at Weill Cornell, a hands-on research program for aspiring young scientists from 2020–2022. In that vein, she spearheaded successful efforts to secure external funding and matching funds from the graduate school to provide stipends to these students during their participation in the program. Gabriella also launched the “Asian Perspectives on the Hidden Curriculum” discussion series, sponsored by the Rockefeller Office of Diversity, Equity, and Inclusion Microgrant Program, which aims to address training inequities and to build a stronger Asian community in the sciences. Finally, she has also engaged actively in numerous science outreach programs sponsored by the New York Academy of Sciences, including as an Afterschool STEM Teacher, a Scientist in Residence, and a Panelist.
 TPCB alumna, Dr. Chloe Burnside commented, “What is most impressive about Gabriella’s commitment to service is the way that she embodies it in everything that she does. From the kind and diligent way that she interacts with and helps her peers, to the community and morale she creates in the groups she forms and leads, to the actions she puts in place to ensure the upholding of her values and ideals. Gabriella works hard in everything she does to make academia a more equitable, safe, and welcoming space, allowing more groups to enjoy and understand science in the way that she does.”
TPCB alumna, Dr. Chloe Burnside commented, “What is most impressive about Gabriella’s commitment to service is the way that she embodies it in everything that she does. From the kind and diligent way that she interacts with and helps her peers, to the community and morale she creates in the groups she forms and leads, to the actions she puts in place to ensure the upholding of her values and ideals. Gabriella works hard in everything she does to make academia a more equitable, safe, and welcoming space, allowing more groups to enjoy and understand science in the way that she does.”
In addition to her exceptional leadership of these programs, Gabriella is also an outstanding scientist, whose research focuses on understanding the biophysical basis for how DNA methylation is read and recognized within hierarchically organized chromosomes, and how these cellular functions impact diseases such as Rett syndrome. She is currently a senior graduate student working with Prof. Shixin Liu at Rockefeller, and she plans to continue onto postdoctoral training after graduation.
The TPCB Student Service Awardee is selected each year by a committee of TPCB students and faculty, chaired by Prof. Daniel Heller at MSK, based on nominations solicited across the program each spring. Gabriella received the award at the annual TPCB Summer Barbeque, which was held on the campus of Rockefeller University on July 23, 2024, and organized by TPCB students Colin Burdette, Nicholas Carbone, and Zirong Chen.

June 2024
Four TPCB Students Awarded NSF Graduate Fellowships
TPCB congratulates our four current and incoming students who have been awarded the prestigious National Science Foundation (NSF) Graduate Research Fellowship! This program recognizes and supports outstanding graduate students across the country in science, technology, engineering, and mathematics. [View the complete list at: NSF]
 • Karl Lin was awarded a fellowship for his project entitled, “Evolving Nanobodies into a Genetically Encoded Multifunctional Tag System”. His project aims to develop miniturized antibodies as genetically encoded, ligandable protein tags for diverse applications such as fluorescence imaging, affinity pulldown, and targeted proteolysis. Karl is currently a third-year graduate student working with Prof. Heeseon An at MSK. Prior to entering TPCB in 2022, he completed his undergraduate training at Middlebury College and a Fulbright Fellowship at the University of Tokyo.
• Karl Lin was awarded a fellowship for his project entitled, “Evolving Nanobodies into a Genetically Encoded Multifunctional Tag System”. His project aims to develop miniturized antibodies as genetically encoded, ligandable protein tags for diverse applications such as fluorescence imaging, affinity pulldown, and targeted proteolysis. Karl is currently a third-year graduate student working with Prof. Heeseon An at MSK. Prior to entering TPCB in 2022, he completed his undergraduate training at Middlebury College and a Fulbright Fellowship at the University of Tokyo.
 • Deepika Nambiar was awarded a fellowship for her project entitled, “Investigating the Mechanism and Machinery Regulating Damaged Ribosomes in Mammalian Cells”. She is studying the biological factors and molecular mechanisms that regulate the potential repair or degradation of damaged ribosomes, which are essential protein factories in the cell. Deepika is currently a third-year graduate student working with Prof. Heeseon An at MSK. Before joining TPCB in 2022, she completed her undergraduate studies at Barnard College.
• Deepika Nambiar was awarded a fellowship for her project entitled, “Investigating the Mechanism and Machinery Regulating Damaged Ribosomes in Mammalian Cells”. She is studying the biological factors and molecular mechanisms that regulate the potential repair or degradation of damaged ribosomes, which are essential protein factories in the cell. Deepika is currently a third-year graduate student working with Prof. Heeseon An at MSK. Before joining TPCB in 2022, she completed her undergraduate studies at Barnard College.
 • Leandro Pimentel Marcelino was awarded a fellowship for his project entitled, “Exploring the Role of Guanosine-5´-triphosphate (GTP) and Capping in γ-Tubulin Ring Complex’s Architecture”. In this proposal, he seeks to understand the role of GTP binding by γ-tubulin in initiating and stabilizing microtubules, a crucial component in cellular transport and cell division. Leandro is currently a third-year graduate student working with Prof. Tarun Kapoor at Rockefeller. He completed his undergraduate studies at City College of New York before joining TPCB in 2022.
• Leandro Pimentel Marcelino was awarded a fellowship for his project entitled, “Exploring the Role of Guanosine-5´-triphosphate (GTP) and Capping in γ-Tubulin Ring Complex’s Architecture”. In this proposal, he seeks to understand the role of GTP binding by γ-tubulin in initiating and stabilizing microtubules, a crucial component in cellular transport and cell division. Leandro is currently a third-year graduate student working with Prof. Tarun Kapoor at Rockefeller. He completed his undergraduate studies at City College of New York before joining TPCB in 2022.
 • Emily Scheib was awarded a fellowship for her proposal entitled, “Characterizing the Evolution of a Cadherin Bond Involved in Sensory Perception”. She envisoned a project to develop structural, evolutionary, and mechanistic insights into two causative genes of Usher syndrome, the leading cause of inherited dual vision and auditory impairment. Emily carried out her undergraduate studies at The Ohio State University in 2023 followed by a research assistantship at the University of Chicago. She joined TPCB in 2024 and is currently pursuing her first-year laboratory rotations.
• Emily Scheib was awarded a fellowship for her proposal entitled, “Characterizing the Evolution of a Cadherin Bond Involved in Sensory Perception”. She envisoned a project to develop structural, evolutionary, and mechanistic insights into two causative genes of Usher syndrome, the leading cause of inherited dual vision and auditory impairment. Emily carried out her undergraduate studies at The Ohio State University in 2023 followed by a research assistantship at the University of Chicago. She joined TPCB in 2024 and is currently pursuing her first-year laboratory rotations.
January 2024
Put A Ring On It: A New Mechanism of Bacterial Immunity to Viruses
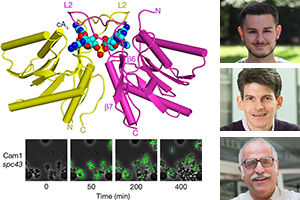 CRISPR-Cas systems are being used extensively in genetic engineering in a wide range of cellular contexts and, most recently, as a new means to treat human genetic diseases. However, these systems originated as an adaptive immune system in bacteria that protects them against viral invaders. Most CRISPR systems mediate this immunity by using RNA to interfere with viral nucleic acids. One of the most widely found systems, type III CRISPR, is not an exception, however, it possesses an additional signaling cascade that enhances its immunity mechanism. Uniquely, the type III CRISPR effector Cas10 cleaves single-stranded DNA and also produces cyclic oligoadenylate molecules from ATP. These cyclic adenylates act as second messengers to activate additional effectors. These effectors have a CARF domain that binds the cyclic oligoadenyalate ligand and are fused to domains that provide additional immunity. Recently, TPCB student Christian Baca and colleagues identified a novel type III CRISPR accessory protein named Cam1, in which the CARF domain is fused to a transmembrane helix not found in other CARF effectors. In a collaborative effort between the labs of TPCB faculty members Prof. Luciano Marraffini at Rockefeller and Prof. Dinshaw Patel at MSK, Cam1 was shown to provide immunity to bacteria against viruses by arresting the infected host cell, making it inhospitable for viral replication, and thus preventing the spread of the virus to neighboring bacteria within the population. Cam1 is a tetrameric protein that forms a pore in the bacterial cell membrane. Upon activation by binding of cyclic tetraadenylate, a Cam1 pore causes depolarization across the bacterial membrane. These findings were published recently in Nature, and are a part of growing body of work showing that CRISPR-Cas systems do not always mediate interference through destruction of nucleic acids and can provide immunity through diverse mechanisms. [Read more at: Nature]
CRISPR-Cas systems are being used extensively in genetic engineering in a wide range of cellular contexts and, most recently, as a new means to treat human genetic diseases. However, these systems originated as an adaptive immune system in bacteria that protects them against viral invaders. Most CRISPR systems mediate this immunity by using RNA to interfere with viral nucleic acids. One of the most widely found systems, type III CRISPR, is not an exception, however, it possesses an additional signaling cascade that enhances its immunity mechanism. Uniquely, the type III CRISPR effector Cas10 cleaves single-stranded DNA and also produces cyclic oligoadenylate molecules from ATP. These cyclic adenylates act as second messengers to activate additional effectors. These effectors have a CARF domain that binds the cyclic oligoadenyalate ligand and are fused to domains that provide additional immunity. Recently, TPCB student Christian Baca and colleagues identified a novel type III CRISPR accessory protein named Cam1, in which the CARF domain is fused to a transmembrane helix not found in other CARF effectors. In a collaborative effort between the labs of TPCB faculty members Prof. Luciano Marraffini at Rockefeller and Prof. Dinshaw Patel at MSK, Cam1 was shown to provide immunity to bacteria against viruses by arresting the infected host cell, making it inhospitable for viral replication, and thus preventing the spread of the virus to neighboring bacteria within the population. Cam1 is a tetrameric protein that forms a pore in the bacterial cell membrane. Upon activation by binding of cyclic tetraadenylate, a Cam1 pore causes depolarization across the bacterial membrane. These findings were published recently in Nature, and are a part of growing body of work showing that CRISPR-Cas systems do not always mediate interference through destruction of nucleic acids and can provide immunity through diverse mechanisms. [Read more at: Nature]
December 2023
Congratulations to TPCB’s 2023 Graduates
TPCB congratulates our 2023 graduates, Adi Berman (Kapoor Lab, RU), Ayala Carl (Alushin Lab, RU), Chen Chen (Heller Lab, MSK), Natalie Jones (Kapoor Lab, RU), Ilana Kotliar (Sakmar Lab, RU), Jordan Mattheisen (Sakmar Lab, RU), and Nicholas Prescott (David Lab, MSK)! The students’ thesis research spanned a wide range of topics, using chemical biology tools to study cytoskeletal dynamics, protein architecture, and novel drug targets. After graduating, they have gone onto postdoctoral training and positions as staff scientists and scientific consultants.
Students who defended their theses in the spring received their PhD degrees at the Weill Cornell Convocation Ceremony on May 18, 2023 or at The Rockefeller University Convocation Ceremony on June 1, 2023. Students in MSK labs were also recognized at the MSK Academic Convocation on May 17, 2023. The remaining graduates will be recognized formally at next year’s ceremonies. We wish our newest alumni all the best as they continue onto the next stages of their careers! [Read more at: Weill Cornell | Rockefeller | Memorial Sloan Kettering]
 • Adi Berman, PhD – “Breaking Down Microtubule Formation: Characterizing the γ-Tubulin Ring Complex”
• Adi Berman, PhD – “Breaking Down Microtubule Formation: Characterizing the γ-Tubulin Ring Complex”
TPCB Mentor: Prof. Tarun Kapoor, The Rockefeller University
Current Position: Postdoctoral Fellow, Regeneron Pharmaceuticals
 • Ayala Carl, PhD – “Visualizing Cytoskeletal Force Transduction”
• Ayala Carl, PhD – “Visualizing Cytoskeletal Force Transduction”
TPCB Mentor: Prof. Gregory Alushin, The Rockefeller University
Current Position: Scientist, Field Applications, Thermo Fisher Scientific
 • Chen Chen, PhD – “Using Nanotechnology to Modulate and Monitor Pharmacokinetics in Drug Development”
• Chen Chen, PhD – “Using Nanotechnology to Modulate and Monitor Pharmacokinetics in Drug Development”
TPCB Mentor: Prof. Daniel Heller, Memorial Sloan Kettering
Current Position: Postdoctoral Fellow, Genentech
 • Natalie Jones, PhD – “Tuning AAA Protein Machines Using Small Molecule Tools”
• Natalie Jones, PhD – “Tuning AAA Protein Machines Using Small Molecule Tools”
TPCB Mentor: Prof. Tarun Kapoor, The Rockefeller University
Current Position: Postdoctoral Fellow, Prof. Leemor Joshua-Tor, CSHL
 • Ilana Kotliar, PhD – “Multiplexed Mapping of the GPCR-RAMP Interactome”
• Ilana Kotliar, PhD – “Multiplexed Mapping of the GPCR-RAMP Interactome”
TPCB Mentor: Prof. Thomas Sakmar, The Rockefeller University
Current Position: Life Sciences Consultant, EY-Parthenon
 • Jordan Mattheisen, PhD – “Bioorthogonal Tethering of Drug Fragments to Engineered GPCRs”
• Jordan Mattheisen, PhD – “Bioorthogonal Tethering of Drug Fragments to Engineered GPCRs”
TPCB Mentor: Prof. Thomas Sakmar, The Rockefeller University
Current Position: Postdoctoral Fellow, AstraZeneca
 • Nicholas Prescott, PhD – “Dissecting the Chromatin Regulation of Hepatitis B Virus”
• Nicholas Prescott, PhD – “Dissecting the Chromatin Regulation of Hepatitis B Virus”
TPCB Mentor: Prof. Yael David, Memorial Sloan Kettering
Current Position: Postdoctoral Fellow, Prof. Hironori Funabiki, The Rockefeller University
September 2023
TPCB Students Enjoy the 2023 Career Development Retreat
TPCB students attended our annual Student Career Development Retreat on September 26–27, 2023 at the scenic Woodloch Resort in New Jersey. The program featured a keynote presentation by Jesse H. Ausubel, Director of the Program for the Human Environment at Rockefeller and an alumni panel discussion with Prof. Vidhya Rangaraju (Group Leader, Max Planck Florida Institute for Neuroscience; TPCB ‘13, Ryan Lab) and Dr. Christopher Stratton (Senior Scientist, Janssen Research and Development; TPCB ‘12, Tan Lab).

Students engaged in some friendly competition in our annual 3-Minute Thesis Talk event, around the go-kart track, and on the volleyball court, and also enjoyed paddleboarding and kayaking around the lake, and exercising their creativity in a painting class. The event was organized by rising 3rd-year students Jared Ramsey, Nicolai Tornow, and Nora Zhou and received generous financial support from the TPCB NIH T32 Chemistry–Biology Interface Training Grant.




August 2023
ChBSP Summer Internship Program Hosts Another Outstanding Group of Undergraduates
 The Tri-Institutional Chemical Biology Summer Program (ChBSP) hosted another group of outstanding undergraduate chemical biologists this summer. The students pursued laboratory research in TPCB labs and attended scientific presentations by TPCB faculty members. The group also interacted regularly with TPCB graduate students who led journal club discussions of recent literature. They also participated in career development training and enjoyed a variety of outings in New York City. The summer culminated on August 9, 2023 in a poster session where the ChBSP interns presented their research alongside students from other summer programs at MSK.
The Tri-Institutional Chemical Biology Summer Program (ChBSP) hosted another group of outstanding undergraduate chemical biologists this summer. The students pursued laboratory research in TPCB labs and attended scientific presentations by TPCB faculty members. The group also interacted regularly with TPCB graduate students who led journal club discussions of recent literature. They also participated in career development training and enjoyed a variety of outings in New York City. The summer culminated on August 9, 2023 in a poster session where the ChBSP interns presented their research alongside students from other summer programs at MSK.
ChBSP is directed by TPCB faculty member Prof. Daniel Bachovchin, and receives generous financial support from the Sanders Tri-Institutional Discovery Institute, led by Dr. Peter Meinke. The program is organized by the MSK Office of Scientific Education and Training, led by Dr. Ushma Neill. The 2023 journal club was led by TPCB student David Hsieh, with Atara Neugroschl, Jared Ramsey, Alexa Strauss, and Charles Warren. Notably, Atara and Charlie are themselves ChBSP alumni!





Judey DaRos, University of Pittsburgh, Class of 2024
Investigating NLRP3 Inflammasome Activation, with Prof. Daniel Bachovchin, MSK
Mahak Kathpalia, University of Wisconsin, Madison, Class of 2024
Characterizing Novel Vulnerabilities Associated with Acquired mTOR-inhibitor Resistance in Breast Cancer, with Prof. John Blenis, Weill Cornell
Kaitliyn Ko, University of Chicago, Class of 2024
RNA May Act as a Peroxidase within Neurons to Cause Certain Neurodegenerative Disorders, with Prof. Samie Jaffrey, Weill Cornell
Raymond Lam, University of Texas, Austin, Class of 2024
Engineering Pyrrolysyl-tRNA Synthetase for Incorporation of Methylated Lysine, with Prof. Minkui Luo, MSK
Dat Nguyen, Oberlin College, Class of 2024
Synthesis and Evaluation of ASPIRe Analogs to Improve On-target Activity of AAA Inhibitors, with Prof. Tarun Kapoor, Rockefeller
Mihika Shah, Stevens Institute of Technology, Class of 2024
Decoding Enhancer Promoter Interactions with Single-molecule Visualization and Manipulation, with Prof. Shixin Liu, Rockefeller
Group photo courtesy of MSK Marketing & Communication.
July 2023
TPCB Students Serve as Guest Editors of Special Issue of ChemBioChem
 TPCB students were honored to guest edit a special issue of ChemBioChem, entitled “Next Generation of Chemical Biology”. Editor-in-Chief, Dr. Ruben Ragg, invited the students to assemble the special issue after attending the 2021 Tri-Institutional Chemical Biology Symposium, with the goal of engaging the next generation of scientists in chemical biology. The project included guest editorials by Nicholas Prescott and Hsin-Che Huang on the unique scientific fluency of chemical biologists across disciplines, by Gabriella Chua, J. Jeya Vandana, and Chun-Cheng Hsieh on best practices in graduate training and mentorship, and by Gianna Stella on the value of failure as a critical stepping stone to scientific and personal growth. TPCB student Lucas Repeta also served as a member of the editorial team. The issue featured primary research articles from across the chemical biology community, and a conference report on the 2022 Proteomics in Cell Biology and Disease Mechanisms by TPCB student Ilana Kotliar.
TPCB students were honored to guest edit a special issue of ChemBioChem, entitled “Next Generation of Chemical Biology”. Editor-in-Chief, Dr. Ruben Ragg, invited the students to assemble the special issue after attending the 2021 Tri-Institutional Chemical Biology Symposium, with the goal of engaging the next generation of scientists in chemical biology. The project included guest editorials by Nicholas Prescott and Hsin-Che Huang on the unique scientific fluency of chemical biologists across disciplines, by Gabriella Chua, J. Jeya Vandana, and Chun-Cheng Hsieh on best practices in graduate training and mentorship, and by Gianna Stella on the value of failure as a critical stepping stone to scientific and personal growth. TPCB student Lucas Repeta also served as a member of the editorial team. The issue featured primary research articles from across the chemical biology community, and a conference report on the 2022 Proteomics in Cell Biology and Disease Mechanisms by TPCB student Ilana Kotliar.
Read the full issue: Next Generation of Chemical Biology
• Prescott, N. A.; Huang, H.-C. “Scientific Fluency as the Greatest Strength of Chemical Biologists.” ChemBioChem, 2023, 24, e202300053.
• Chua, G. N. L.; Vandana, J. J.; Hsieh, C.-C. “Students’ Perspective on Scientific Training.” ChemBioChem, 2023, 24, e202300054.
• Stella, G. “Sharing Failure as a Graduate Student.” ChemBioChem, 2023, 24, e202300104.
ChBSP Summer Program Welcomes 2023 Chemical Biology Interns
 TPCB welcomes this year’s summer undergraduate interns in the Chemical Biology Summer Program (ChBSP)! Now in its sixth year, the program provides students with unique experience in chemical biology research in TPCB labs. Students also participate in faculty seminars, professional development seminars, and a journal club hosted by current TPCB graduate students. Alumni of the program have continued onto PhD training in a wide range of prestigious graduate programs, including TPCB itself! The program will culimate in a poster session on August 9, 2023 hosted at MSK. The program is directed by TPCB faculty member Prof. Daniel Bachovchin and Dr. Ushma Neill, who leads the MSK Office of Scientific Education & Training. Generous financial support is provided by the Sanders Innovation & Education Initiative of the Tri-Institutional Therapeutics Discovery Institute. [Read more]
TPCB welcomes this year’s summer undergraduate interns in the Chemical Biology Summer Program (ChBSP)! Now in its sixth year, the program provides students with unique experience in chemical biology research in TPCB labs. Students also participate in faculty seminars, professional development seminars, and a journal club hosted by current TPCB graduate students. Alumni of the program have continued onto PhD training in a wide range of prestigious graduate programs, including TPCB itself! The program will culimate in a poster session on August 9, 2023 hosted at MSK. The program is directed by TPCB faculty member Prof. Daniel Bachovchin and Dr. Ushma Neill, who leads the MSK Office of Scientific Education & Training. Generous financial support is provided by the Sanders Innovation & Education Initiative of the Tri-Institutional Therapeutics Discovery Institute. [Read more]
This year’s ChBSP summer interns are:
- • Judey DaRos, University of Pittsburgh, Class of 2024, Chemistry
(with Prof. Daniel Bachovchin, Sloan Kettering) - • Mahak Kathpalia, U. Wisconsin-Madison, Class of 2024, Chemistry / Cellular & Mol. Biol.
(with Prof. John Blenis, Weill Cornell) - • Kaitlyn Ko, University of Chicago, Class of 2024, Chemistry
(with Prof. Samie Jaffrey, Weill Cornell) - • Raymond Lam, University of Texas at Austin, Class of 2024, Chemistry
(with Prof. Minkui Luo, Sloan Kettering) - • Dat Nguyen, Oberlin College, Class of 2024, Biochemistry / Chemistry
(with Prof. Tarun Kapoor, Rockefeller) - • Mihika Shah, Stevens Institute of Technology, Class of 2024, Chemical Biology
(with Prof. Shixin Liu, Rockefeller)
June 2023
Gianna Stella Recognized with Inaugural TPCB Service Award
 TPCB has a strong culture of service, and our students engage in numerous science outreach, peer mentoring, and scientific training programs, across the Tri-Institutional campuses and beyond. To recognize these important contributions, the program recently established the TPCB Student Service Award, to be awarded annually to a student who has undertaken exceptional service to the scientific community. Recently, Gianna Stella was named the inaugural recipient of the award for 2023. Gianna co-chairs the Tri-Institutional Outreach Committee and co-founded the Tri-Institutional Mentorship Initiative (TIMI) in 2020. TIMI provides mentorship to undergraduate and postgraduate students who are applying to PhD programs in the biomedical sciences, with a particular focus on students from underserved backgrounds. In three short years, this grassroots program has grown to 79 mentor–mentee pairs and has recruited graduate student mentors from every PhD program across the three institutions. TIMI has had a remarkable impact, reaching mentees from across the U.S. and 13 countries around the world this year, with 96% of the U.S. students receiving at least one admission offer.
TPCB has a strong culture of service, and our students engage in numerous science outreach, peer mentoring, and scientific training programs, across the Tri-Institutional campuses and beyond. To recognize these important contributions, the program recently established the TPCB Student Service Award, to be awarded annually to a student who has undertaken exceptional service to the scientific community. Recently, Gianna Stella was named the inaugural recipient of the award for 2023. Gianna co-chairs the Tri-Institutional Outreach Committee and co-founded the Tri-Institutional Mentorship Initiative (TIMI) in 2020. TIMI provides mentorship to undergraduate and postgraduate students who are applying to PhD programs in the biomedical sciences, with a particular focus on students from underserved backgrounds. In three short years, this grassroots program has grown to 79 mentor–mentee pairs and has recruited graduate student mentors from every PhD program across the three institutions. TIMI has had a remarkable impact, reaching mentees from across the U.S. and 13 countries around the world this year, with 96% of the U.S. students receiving at least one admission offer.
In addition to her work on TIMI, Gianna was recognized by her peers for her steadfast commitment to representing graduate student interests and concerns to institutional leadership. In her roles as an elected student representative at both Weill Cornell and Rockefeller, she has advocated successfully for increased mental health support services and new diversity, equity, and inclusion programs. Gianna is currently a graduate student working with Prof. Luciano Marraffini at Rockefeller investigating the molecular mechanisms of CRISPR-based immunity in bacteria. Prof. Marraffini commented, “Gianna has demonstrated exemplary leadership skills and a devotion to improving the lives of people, not only within the university, but also the community at large. Her dedication to teaching and improving scientific literacy in the community is awe-inspiring.” Nominations and supporting letters were solicited in March and April of this year and reviewed by a committee of TPCB students and faculty, chaired by Prof. Daniel Heller of MSK. [Read more about: TIMI]
May 2023
Arvin Dar, Jiankun Lyu, Tobias Meyer, and Michael Rout join the TPCB Faculty
Four new faculty members have recently joined TPCB, bringing world-class research expertise and training opportunities in small-molecule drug discovery and the molecular underpinnings of cellular functions.
 • Prof. Arvin Dar joined the Sloan Kettering faculty in the Chemical Biology Program in June 2023. His lab explores cancer mechanisms and new paradigms in drug discovery by integrating approaches in chemistry, biochemistry, structure, and genetics. In particular, his work focuses on understanding and modulating the molecular mechanisms of kinase function in cancer. Prof. Dar. completed his PhD studies at the University of Toronto and postdoctoral training at UCSF before beginning his independent career at the Mount Sinai School of Medicine. [Read more]
• Prof. Arvin Dar joined the Sloan Kettering faculty in the Chemical Biology Program in June 2023. His lab explores cancer mechanisms and new paradigms in drug discovery by integrating approaches in chemistry, biochemistry, structure, and genetics. In particular, his work focuses on understanding and modulating the molecular mechanisms of kinase function in cancer. Prof. Dar. completed his PhD studies at the University of Toronto and postdoctoral training at UCSF before beginning his independent career at the Mount Sinai School of Medicine. [Read more]
 • Prof. Jiankun Lyu recently joined the faculty at Rockefeller as head of the Evnin Family Laboratory of Computational Molecular Discovery in January 2023. His lab is developing powerful new computational approaches for molecular docking of massive libraries to accelerate drug discovery. By leveraging “make-on-demand” virtual libraries, they seek to discover novel, potent, and selective ligands for new biological targets. Prof. Lyu carried out his PhD studies at East China University of Science & Technology, in collaboration with Prof. Brian Shoichet at UCSF, whose lab he then joined for his postdoctoral training. [Read more]
• Prof. Jiankun Lyu recently joined the faculty at Rockefeller as head of the Evnin Family Laboratory of Computational Molecular Discovery in January 2023. His lab is developing powerful new computational approaches for molecular docking of massive libraries to accelerate drug discovery. By leveraging “make-on-demand” virtual libraries, they seek to discover novel, potent, and selective ligands for new biological targets. Prof. Lyu carried out his PhD studies at East China University of Science & Technology, in collaboration with Prof. Brian Shoichet at UCSF, whose lab he then joined for his postdoctoral training. [Read more]
 • Prof. Tobias Meyer is a leader in the development of chemical biology tools to study intracellular signaling processes that control critical cellular functions such as cell migration, proliferation, and senescence, using cellular and mouse models. He completed his PhD training at the Biozentrum of the University of Basel in Switzerland, then came to the U.S. for postdoctoral training with Prof. Lubert Stryer at Stanford. He began his independent career at the Duke University Medical Center before moving to Stanford in 2000, where he served as Chair of the Department of Chemical and Systems Biology from 2011 to 2016. He was recruited to Weill Cornell Medicine in 2020 as the Joseph C. Hinsey Professor of Biochemistry. [Read more]
• Prof. Tobias Meyer is a leader in the development of chemical biology tools to study intracellular signaling processes that control critical cellular functions such as cell migration, proliferation, and senescence, using cellular and mouse models. He completed his PhD training at the Biozentrum of the University of Basel in Switzerland, then came to the U.S. for postdoctoral training with Prof. Lubert Stryer at Stanford. He began his independent career at the Duke University Medical Center before moving to Stanford in 2000, where he served as Chair of the Department of Chemical and Systems Biology from 2011 to 2016. He was recruited to Weill Cornell Medicine in 2020 as the Joseph C. Hinsey Professor of Biochemistry. [Read more]
 • Prof. Michael Rout studies the molecular architecture of macromolecular complexes with a particular focus on the nuclear pore complex (NPC). His lab leverages a combination of approaches including proteomics, interactomics, and cryo-electron microscopy to understand how the proteins that comprise the NPC interact, contribute to overall structure, and control passage of molecules into and out of the nucleus. He also leads the NIH-funded National Center for Dynamic Interactome Research. Prof. Rout carried out his scientific training at the University of Cambridge and Rockefeller and joined the Rockefeller faculty in 1997. [Read more]
• Prof. Michael Rout studies the molecular architecture of macromolecular complexes with a particular focus on the nuclear pore complex (NPC). His lab leverages a combination of approaches including proteomics, interactomics, and cryo-electron microscopy to understand how the proteins that comprise the NPC interact, contribute to overall structure, and control passage of molecules into and out of the nucleus. He also leads the NIH-funded National Center for Dynamic Interactome Research. Prof. Rout carried out his scientific training at the University of Cambridge and Rockefeller and joined the Rockefeller faculty in 1997. [Read more]
April 2023
TPCB Students Recognized with NSF Graduate Fellowships
TPCB is delighted to share that two of our students have been awarded fellowships by the National Science Foundation (NSF). These prestigious graduate research fellowships provide three years of support during training, and recognize outstanding students across the country in science, technology, engineering, and mathematics. [View the complete list at: NSF]
 • Victoria Jordan was awarded a fellowship for her project entitled, “Studying Ubiquitin Ser57 Phosphorylation as a Regulator of Neuronal Differentiation”. The proposal involves developing a transition-state probe to study phosphorylation of ubiquitin and the potential role of this post-translational modification in neuronal differentiation. Victoria is currently a third-year graduate student working with Prof. Heeseon An at MSK. She completed her undergraduate training at Boston University and joined TPCB in 2021.
• Victoria Jordan was awarded a fellowship for her project entitled, “Studying Ubiquitin Ser57 Phosphorylation as a Regulator of Neuronal Differentiation”. The proposal involves developing a transition-state probe to study phosphorylation of ubiquitin and the potential role of this post-translational modification in neuronal differentiation. Victoria is currently a third-year graduate student working with Prof. Heeseon An at MSK. She completed her undergraduate training at Boston University and joined TPCB in 2021.
 • Christopher Nieves Escobar has been recognized with a fellowship for his project entitled, “Photoredox-Catalyzed Synthesis of 3-Oxetanols and 3-Azetidinols from Carboxylic Acids”. He aims to develop novel syntheses of oxetane-containing bioisosteres of carboxylic acid-based drugs to provide analogues with improved pharmacological properties. Chris is currently a third-year graduate student in the lab of Prof. Derek Tan at MSK. He carried out his undergraduate training at the University of Puerto Rico, Humacao and joined TPCB in 2021.
• Christopher Nieves Escobar has been recognized with a fellowship for his project entitled, “Photoredox-Catalyzed Synthesis of 3-Oxetanols and 3-Azetidinols from Carboxylic Acids”. He aims to develop novel syntheses of oxetane-containing bioisosteres of carboxylic acid-based drugs to provide analogues with improved pharmacological properties. Chris is currently a third-year graduate student in the lab of Prof. Derek Tan at MSK. He carried out his undergraduate training at the University of Puerto Rico, Humacao and joined TPCB in 2021.
December 2022
Congratulations to TPCB’s 2022 Graduates
TPCB congratulates our 2022 graduates, Tandrila Das (Hang Lab, RU), Jakob Hebert (David Lab, MSK), Mizuho Horioka (Sakmar Lab, RU), Igor Maksimovic (David Lab, MSK), Elizabeth Orth-He (Bachovchin Lab, MSK), Devin Ray (David Lab, MSK), and Emily Rundlet (Blanchard Lab, WCM)! The students conducted thesis research across the spectrum of chemical biology, studying small-molecule enzyme inhibitors as biological probes, the impacts of protein post-translational modifications on cellular functions, protein structural dynamics in fundamental cellular processes, and the roles of G-protein coupled receptor mutations in disease. They have continued on to postdoctoral training, staff scientist positions, and scientific consulting careers.
Students who defended their theses in the spring received their PhD degrees at the Weill Cornell Convocation Ceremony on May 18, 2022 or at The Rockefeller University Convocation Ceremony on June 9, 2022. Students in MSK labs were also recognized at the MSK Academic Convocation on May 18, 2022. The remaining graduates will be recognized formally at next year’s ceremonies. We wish our newest alumni all the best as they continue onto the next stages of their careers! [Read more at: Weill Cornell | Rockefeller | Memorial Sloan Kettering]
 • Tandrila Das, PhD – “Functional Analysis of S-Palmitoylated IFITM3 Antiviral Activity and Regulation”
• Tandrila Das, PhD – “Functional Analysis of S-Palmitoylated IFITM3 Antiviral Activity and Regulation”
TPCB Mentor: Prof. Howard Hang, The Rockefeller University
Current Position: Scientist, Vividion Therapeutics
 • Jakob Hebert, PhD – “The SWI/SNF-related Protein SMARCA3 is a Histone H3K23 Ubiquitin Ligase that Regulates H3K9me3 in Cancer”
• Jakob Hebert, PhD – “The SWI/SNF-related Protein SMARCA3 is a Histone H3K23 Ubiquitin Ligase that Regulates H3K9me3 in Cancer”
TPCB Mentor: Prof. Yael David, Memorial Sloan Kettering
Current Position: Senior Scientist, Cymba X
 • Mizuho Horioka, PhD – “Activity Profiling of Genetic Variants of the CYSLTR2 Oncogene Responsible for Uveal Melanoma”
• Mizuho Horioka, PhD – “Activity Profiling of Genetic Variants of the CYSLTR2 Oncogene Responsible for Uveal Melanoma”
TPCB Mentor: Prof. Thomas Sakmar, The Rockefeller University
Current Position: Scientist, Exscientia
 • Igor Maksimovic, PhD – “A Chemical Toolbox to Study Histone Glycation”
• Igor Maksimovic, PhD – “A Chemical Toolbox to Study Histone Glycation”
TPCB Mentor: Prof. Yael David, Memorial Sloan Kettering
Current Position: Consultant, Boston Consulting Group
 • Elizabeth Orth-He, PhD – “Aminopeptidase Inhibition Activates the CARD8 and NLRP1 Inflammasomes”
• Elizabeth Orth-He, PhD – “Aminopeptidase Inhibition Activates the CARD8 and NLRP1 Inflammasomes”
TPCB Mentor: Prof. Daniel Bachovchin, Memorial Sloan Kettering
Current Position: Consultant, McKinsey & Co
 • Devin Ray, PhD – “Development of Chemical Tools to Investigate and Manipulate Cellular Processes”
• Devin Ray, PhD – “Development of Chemical Tools to Investigate and Manipulate Cellular Processes”
TPCB Mentor: Prof. Yael David, Memorial Sloan Kettering
Current Position: MD-PhD Student, Tri-Institutional MD-PhD Program
 • Emily Rundlet, PhD – “Structural Dynamics of tRNA Translocation Through the Ribosome During Protein Synthesis”
• Emily Rundlet, PhD – “Structural Dynamics of tRNA Translocation Through the Ribosome During Protein Synthesis”
TPCB Mentor: Prof. Scott Blanchard, Weill Cornell Medicine
Current Position: Postdoctoral Fellow, Prof. Scott Blanchard, St. Jude Children’s Research Hospital
September 2022
Chemical Biology Showcased at 18th Annual Tri-I Symposium
The 18th Annual Tri-Institutional Chemical Biology Symposium was hosted by TPCB on September 8, 2022 on the campus of The Rockefeller University. The event was held in a hybrid format and attended both in person and online by some 170 scientists from around the world. The event featured keynote seminars by leading faculty in the field of chemical biology, short talks by several TPCB students, and poster presentations from labs across seven institutions.


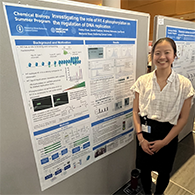
[First photo]: Participants attend Prof. Angela Koehler’s keynote seminar. [Second photo]: TPCB student Gabriella Chua presents a short talk on her research.
Prof. Angela Koehler of the Koch Institute at MIT kicked off the symposium with a seminar on the use of small-molecule microarrays to discover novel probes that address challenging targets including intrinsically disordered proteins, dimeric transcription factors, and hormone receptor splice variants. TPCB student Adi Berman, from the lab of Prof. Tarun Kapoor at Rockefeller, presented her investigations of the role of the γ-tubulin ring complex in regulating microtubule dynamics. Next, TPCB student Gabriella Chua, who works with Prof. Shixin Liu at Rockefeller, spoke about her fluorescence imaging studies of MeCP2, a protein that recognizes methylated DNA and interacts with linker histone H1 to condense chromatin structure. Prof. Simon Scheuring, a TPCB faculty member at Weill Cornell Medicine, capped off the morning session with a lecture on his lab’s use of high-speed atomic force microscopy to assess conformational changes in membrane proteins in response to factors such as temperature, force, and protein–protein interactions.


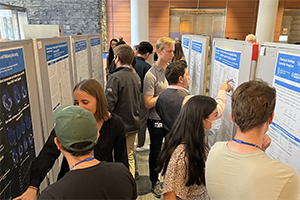
[First photo]: The poster session featured 45 presentations representing labs across 7 institutions. [Second photo]: TPCB student Karl Lin presents a poster on his Fulbright research at the University of Tokyo.
The lunchtime poster session featured presentations by 45 students and postdocs spanning the spectrum of chemical biology. These included efforts to develop new synthetic methods to access bioactive molecules, to discover new natural products from the gut microbiome, to design new inhibitors and PROTACs as potential therapeutics, to develop new platforms to enable target identification for small-molecule probes, to use chemical genetic probes to study protein function, to investigate the structures of large protein assembles, and to carry out computational modeling of biomolecules. Poster presentations were judged by the visiting keynote speakers and TPCB faculty members. Awards comprised of gift cards and journal subscriptions sponsored by TPCB and our promotional partners were given in four categories: Early Stage Students, TPCB Graduate Students, Open Graduate Students, and Postdoctoral Fellows.


[First photo]: TPCB student Ilana Kotliar presents a poster on her research with Prof. Tom Sakmar at Rockefeller. [Second photo]: TPCB student Yuanhuang Chen presents a poster on his master’s research at the University of Michigan.
The afternoon talks began with a faculty keynote lecture by Prof. Heather Pinkett of Northwestern University on the molecular mechanisms ABC transporters, which leveraged biochemical and structural studies to showcase transporter diversity. TPCB student Nicholas Prescott, from Prof. Yael David’s lab at MSK, next shared his work investigating the interplay between chromatin assembly on covalently closed circular DNA in hepatitis B virus and transcription of the viral oncogene HBx. The final keynote presentation was given by Prof. Matthew Bogyo of Stanford University, on the use of chemical probes to study how proteases regulate the gut microbiome.


[First photo]: TPCB student Nicholas Prescott presents a short talk on his research with Prof. Yael David. [Second photo]: Prof. Matthew Bogyo presents a faculty keyone lecture on his research in chemical biology.
The event concluded with presentation of poster awards and a reception for all symposium participants, which was followed by dinner at a local restaurant attended by TPCB students and the faculty speakers.
This year’s symposium was organized by TPCB students Abigail Lemmon and Tiffany Zhang, assisted by long-time TPCB faculty mentor Prof. Daniel Heller of MSK, TPCB Program Coordinator Margie Hinonangan-Mendoza, and TPCB Director Prof. Derek Tan. Seminars were webcast with support from Rockefeller University. Generous promotional support was provided by ACS Chemical Biology, Biochemistry, Cell Chemical Biology, ChemBioChem, Chemical Science, Nature Chemical Biology, Organic & Biomolecular Chemistry, RSC Chemical Biology, and the New York Academy of Sciences.


[First photo]: Symposium speakers and organizers (from left): TPCB Director Prof. Derek Tan, Gabriella Chua, Nicholas Prescott, Adi Berman, Prof. Heather Pinkett, Prof. Matthew Bogyo, Abigail Lemmon, Tiffany Zhang, TPCB Mentor Prof. Daniel Heller. [Second photo]: Winners of the Early Stage Student Poster Awards (from left): Karl Lin, Victoria Jordan, TPCB Director Prof. Derek Tan, Noah Yardeny.
Poster Award Winners
Early Stage Students
• Karl Lin, University of Tokyo (Robert Campbell Lab)
– Towards the Development of Single Fluorescent Protein-based Biosensors for Phosphorylated Intermediates in Central Carbon Metabolism
– ChemBioChem Prize
• Noah Yardeny, TPCB (Geri Lab, Weill Cornell)
– High-throughput Target ID of Native Biomolecules
– Nature Chemical Biology Prize
• Victoria Jordan, TPCB (An Lab, MSK)
– Developing Ubiquitin-ATP Probes to Identify Novel Kinases
– Nature Chemical Biology Prize
TPCB Graduate Students
• Lauren Vostal, TPCB (Kapoor Lab, Rockefeller)
– Analyzing Site-specific and Direct Interactions of AAA Proteins with Cofactors and Substrates in Living Cells
– ChemBioChem Prize
• Ilana Kotliar, TPCB (Sakmar Lab, Rockefeller)
– Itch receptor MRGPRX4 is Differentially Regulated by the Receptor Activity-Modifying Proteins (RAMPs)
– RSC Chemical Biology Prize
• Dominic Rufa, TPCB (Chodera Lab, MSK)
– Towards Chemical Accuracy for Alchemical Free Energy Calculations with Hybrid Physics-based Machine Learning/Molecular Mechanics Potentials
– Cell Chemical Biology Prize
• Ayala Carl, TPCB (Alushin Lab, Rockefeller)
– F-actin Acts as Tension Sensor via Superhelix Structure
– Chemical Science Prize


[First photo]: Winners of the TPCB Graduate Student Poster Awards (from left): Lauren Vostal, Ilana Kotliar, TPCB Director Prof. Derek Tan, Dominic Rufa, Ayala Carl. [Second photo]: Winners of the Open Graduate Student Poster Awards (from left): Avelyn Mae Delos Reyes, Derek Tan, Ifé Akano.
Open Graduate Students
• Vinay Kumar Sapuru, Weill Cornell PBSP (Hite Lab, MSK)
– Structural Titration of Human Inositol Trisphosphate Receptor Reveals Mechanisms of Ligand-dependent Activation and Inhibition
– ChemBioChem Prize
• Avelyn Mae Delos Reyes, Weill Cornell Pharmacology (Tan Lab, MSK)
– Direct Conversion of Carboxylic Acids to 3-Oxetanol Bioisosteres via Photoredox Catalysis
– Nature Chemical Biology Prize
• Ifé Akano, Weill Cornell Pharmacology (David Lab, MSK)
– SMARCA3 is a Novel H3K23 Ubiquitin Ligase that Regulates H3K9me3 in Cancer
– RSC Chemical Biology Prize
Postdoctoral Fellows
• Guoqing Xiang, PhD, Weill Cornell Medicine (Levitz Lab)
– Control of Gaq Signaling Dynamics and GPCR Crosstalk by GRKs
– Cell Chemical Biology Prize
• Lingling Cheng, PhD, MSK (Pertsinidis Lab)
– Revealing Promoter–Enhancer Communication Mechanisms by High-resolution Single-gene Imaging
– Organic & Biomolecular Chemistry Prize
August 2022
ChBSP Summer Interns Pursue Forefront Chemical Biology Research in TPCB Labs
 This summer saw another class of outstanding undergraduate researchers join TPCB labs under the auspices of the Tri-Institutional Chemical Biology Summer Program (ChBSP). This year’s interns pursued research across the spectrum of chemical biology. They also participated in a weekly journal club organized by TPCB graduate students, attended seminars by TPCB faculty members, and participated in professional development workshops. The interns also took some time out to enjoy New York City, including a dinner cruise on the Spirit of New York. The students presented their impressive work at a poster session on August 11, 2022, alongside other interns from Tri-Institutional and MSK summer programs.
This summer saw another class of outstanding undergraduate researchers join TPCB labs under the auspices of the Tri-Institutional Chemical Biology Summer Program (ChBSP). This year’s interns pursued research across the spectrum of chemical biology. They also participated in a weekly journal club organized by TPCB graduate students, attended seminars by TPCB faculty members, and participated in professional development workshops. The interns also took some time out to enjoy New York City, including a dinner cruise on the Spirit of New York. The students presented their impressive work at a poster session on August 11, 2022, alongside other interns from Tri-Institutional and MSK summer programs.





 Casey Chan
Casey Chan
Brown University, Class of 2023
Investigating the Role of H1.4 Phosphorylation on the Regulation of DNA Replication
with Prof. Yael David, MSK
 Isabelle Edmondson
Isabelle Edmondson
Middlebury College, Class of 2023
Thioredoxin Stabilizes the NLRP1 Inflammasome via an ATP-Dependent Inhibitory Association
with Prof. Daniel Bachovchin, MSK
 José Luis Llera Muñoz
José Luis Llera Muñoz
University of Puerto Rico, Rio Piedras, Class of 2023
Screening of Mutations on the Ligand Binding Pocket of an Acyclovir Binding RNA Aptamer
with Prof. Samie Jaffrey, Weill Cornell
 Elizabeth Mari
Elizabeth Mari
Haverford College, Class of 2023
Synthesis of Bioorthogonal Photocatalysts for Proximity Labeling Applications
with Prof. Jacob Geri, Weill Cornell
 Hans Oh
Hans Oh
University of North Carolina, Chapel Hill, Class of 2023
Synthesis toward Chemical Probes of PPA, a Covalent p97 Inhibitor, to Investigate AAA Proteins
with Prof. Tarun Kapoor, Rockefeller
 Sarah Xi
Sarah Xi
Columbia University, Class of 2023
Small Molecule Induction of a Unique T cell Phenotype
with Prof. Ekaterina Vinogradova, Rockefeller
 ChBSP is led by program director Prof. Daniel Bachovchin, and is offered in partnership between TPCB and the MSK Office of Scientific Education & Training, led by Dr. Ushma Neill. The ChBSP journal club was organized by TPCB graduate students David Hsieh and Natalie Jones, and mentored by Gabriella Chua, Victoria Jordan, Ilana Kotliar, Abigail Lemmon, Alexander Payne, Nicholas Prescott, Jared Ramsey, Gianna Stella, Nicole Walker, and Charles Warren. The program receives generous financial support for the interns from the Sanders Innovation and Education Initiative of the Tri-Institutional Therapeutics Discovery Institute, which is led by Dr. Peter Meinke. [Read more]
ChBSP is led by program director Prof. Daniel Bachovchin, and is offered in partnership between TPCB and the MSK Office of Scientific Education & Training, led by Dr. Ushma Neill. The ChBSP journal club was organized by TPCB graduate students David Hsieh and Natalie Jones, and mentored by Gabriella Chua, Victoria Jordan, Ilana Kotliar, Abigail Lemmon, Alexander Payne, Nicholas Prescott, Jared Ramsey, Gianna Stella, Nicole Walker, and Charles Warren. The program receives generous financial support for the interns from the Sanders Innovation and Education Initiative of the Tri-Institutional Therapeutics Discovery Institute, which is led by Dr. Peter Meinke. [Read more]
June 2022
TPCB Students Participate in 2022 NYAS Chemical Biology Symposium
The annual New York Academy of Sciences Chemical Biology Symposium returned live and in-person on May 26, 2022, after being convened online for the last two years. The event was held at Columbia University, and attracted students, faculty, and industry scientists from across NYC and surrounding areas. The program showcased research across the full spectrum of chemical biology and TPCB was well represented, with Lauren Vostal of Prof. Tarun Kapoor’s lab at Rockefeller selected by the organizing committee to give a short talk on her research.
The program launched with a keynote address by Dr. Ayesha Sitlani of the International AIDS Vaccine Initiative (AVI), who discussed the use of monoclonal antibodies to treat infectious diseases. The morning session continued with presentations small-molecule proximity labeling by Dann Parker of Merck, click chemistry-based kinase profiling by Dr. Saundra Denise Field of Pfizer, antibody-drug conjugate development by Dr. Amy Q. Han of Regeneron, and protein-stabilizing therapeutics by Prof. Henry Colecraft of Columbia. After a networking lunch, the afternoon session kicked off with a second keynote address by Prof. Peter Schultz of Scripps, who shared an overview of his lab’s long-standing program in non-natural amino acid incorporation into proteins and organisms, as well as recent work in expanding the genetic code.
 Student presentations were selected from poster abstracts by the organizing committee, and started with a short talk by Amirhossein Jafariyan, a student working with Prof. Virginia Cornish at Columbia on the development of live-yeast hydrogels that secrete wound healing proteins to treat diabetic ulcers. Cassandra Chartier of Prof. Neel Shah’s lab at Columbia then discussed her discoveries surrounding activation of the PTP1B phosphatase by the adaptor protein Grb2. Hoang Anh Phan of the University of Pennsylvania presented her work with Prof. James Petersson on the development of thioamide-stabilized peptides as inhibitors of cathepsin L. Jonathan Kwok, representing Prof. Paramjit Arora’s lab at NYU, shared his research in developing cross-linked helical fork peptides as sequence-specific RNA binders. Lauren Vostal, of TPCB and Prof. Tarun Kapoor’s lab at Rockefeller, spoke about her studies of protein–protein interactions of the AAA protein VCP leveraging amber suppression and photocrosslinking. Finally, Dr. Nivea Pereira De Sa from Prof. Michael Aviola’s lab at Stony Brook presented her structural studies of the sterol glyocsyltransferase SGL1 as a novel target to treat Cryptococcus neoformans fungal infections.
Student presentations were selected from poster abstracts by the organizing committee, and started with a short talk by Amirhossein Jafariyan, a student working with Prof. Virginia Cornish at Columbia on the development of live-yeast hydrogels that secrete wound healing proteins to treat diabetic ulcers. Cassandra Chartier of Prof. Neel Shah’s lab at Columbia then discussed her discoveries surrounding activation of the PTP1B phosphatase by the adaptor protein Grb2. Hoang Anh Phan of the University of Pennsylvania presented her work with Prof. James Petersson on the development of thioamide-stabilized peptides as inhibitors of cathepsin L. Jonathan Kwok, representing Prof. Paramjit Arora’s lab at NYU, shared his research in developing cross-linked helical fork peptides as sequence-specific RNA binders. Lauren Vostal, of TPCB and Prof. Tarun Kapoor’s lab at Rockefeller, spoke about her studies of protein–protein interactions of the AAA protein VCP leveraging amber suppression and photocrosslinking. Finally, Dr. Nivea Pereira De Sa from Prof. Michael Aviola’s lab at Stony Brook presented her structural studies of the sterol glyocsyltransferase SGL1 as a novel target to treat Cryptococcus neoformans fungal infections.
The day culminated with a vibrant poster session presented by students and postdocs from many institutions around the NYC area, including four other states. Poster prizes were sponsored by the New York Academy of Sciences:
• Aweon Richards, New York University (Prof. Tania Lupoli lab)
– An Allosteric Inhibitor of Bacterial Hsp70 Chaperone Mitigates Antibiotic Resistance
• Michael Mohsen, PhD, Yale University (Prof. Ronald Breaker lab)
– Exploiting Natural Riboswitches as Platforms for Evolution and Validation of Synthetic Aptamers
• Yacoba Vroom Minnow, Albert Einstein College of Medicine (Prof. Vern Schramm lab)
– Transition State Analogue Inhibitors of Plasmodium falciparum Hypoxanthine-guanine-xanthine Phosphoribosyltransferase
May 2022
TPCB Students Organize Annual Career Development Retreat
After a three-year hiatus due to the pandemic, TPCB was excited to resume our annual Student Career Development Retreat. The event was held at the Woodloch Resort in the Poconos on May 4-5, 2022 and was organized by second-year students Clare Cahir and Victoria Rasmussen. The program featured a keynote presentation by Dr. Ruth Gotian, Chief Learning Officer at Weill Cornell Medicine and author of award-winning book “The Success Factor”. Students also participated in a panel discussion with TPCB alumni Dr. Paul Dossa,’13, currently an Associate at Venrock, and Dr. Elizabeth George Cisar, ’09, a patent agent who has worked at several firms. The students challenged themselves to present their research in a single static slide in a 3-Minute Thesis Talk competition. Always a highlight of the year, the students also enjoyed social events and activities, including painting, go-kart races, and mini golf. Financial support for this annual program event is provided by TPCB’s NIH T32 Chemistry–Biology Interface Training Grant.



[1] Dr. Ruth Gotian gave the keynote lecture on “The Success Factor”. [2] TPCB alumni Beth George Cisar (left) and Paul Dossa (right) shared their experiences with current students. [3] Jared Ramsey and Charlie Warren hit the go-kart track. [4] (from left): Hsin-Che Huang, Adi Berman, Lauren Vostal, Gianna Stella, Ilana Kotliar, and Adam Rosenzweig show off their paintings.
Olga Boudker Elected to National Academy of Sciences
 TPCB faculty member Prof. Olga Boudker has been elected to the National Academy of Sciences. Academy membership is one of the highest honors in science. Prof. Boudker is recognized for her seminal work in understanding the molecular mechanisms of glutamate transporters in neurons. These cellular machines help transmit signals through nerves by pumping glutamate, a small-molecule neurotransmitter, from cell–cell synapses into brain cells. In this way, they terminate signaling between neurons, help recycle the transmitter, and allow multiple rounds of signaling. This neurotransmission in the brain is the basis for learning, memory, and cognition. Prof. Boudker’s lab at Weill Cornell has hosted numerous TPCB graduate students, and she serves on the TPCB Admissions Committee. She is also an Investigator of the Howard Hughes Medical Institute, as well as acting Chair of the Department of Physiology and Biophysics at Weill Cornell Medicine. Prof. Boudker joins 9 other current TPCB faculty members who have been recognized with this high honor. [ Read more at: Weill Cornell News | National Academy of Sciences ]
TPCB faculty member Prof. Olga Boudker has been elected to the National Academy of Sciences. Academy membership is one of the highest honors in science. Prof. Boudker is recognized for her seminal work in understanding the molecular mechanisms of glutamate transporters in neurons. These cellular machines help transmit signals through nerves by pumping glutamate, a small-molecule neurotransmitter, from cell–cell synapses into brain cells. In this way, they terminate signaling between neurons, help recycle the transmitter, and allow multiple rounds of signaling. This neurotransmission in the brain is the basis for learning, memory, and cognition. Prof. Boudker’s lab at Weill Cornell has hosted numerous TPCB graduate students, and she serves on the TPCB Admissions Committee. She is also an Investigator of the Howard Hughes Medical Institute, as well as acting Chair of the Department of Physiology and Biophysics at Weill Cornell Medicine. Prof. Boudker joins 9 other current TPCB faculty members who have been recognized with this high honor. [ Read more at: Weill Cornell News | National Academy of Sciences ]
April 2022
Entering A New Phase: Linker Histone H1 Orchestrates Genome Organization by Phase Separation with DNA
 Each cell in the body contains some two meters of DNA. To compact this genetic material into the cell nucleus, a group of five proteins called histones collaborate to organize the DNA into a condensed yet readable format. Histone H1, also known as linker histone, has for many years been known to act in concert with the other four histone proteins to compact double-stranded genomic DNA. Together, these histone proteins pack away the vast majority of the genome that eventually form chromosomes as we know them. However, in a new report in Nature Structural and Molecular Biology, a collaborative TPCB team has discovered an unexpected and independent function for linker histone H1. The work was led by TPCB students Dr. Rachel Leicher, Dr. Adewola Osunsade, and Gabriella Chua, and postdoctoral fellow Dr. Sarah Faulkner, along with other colleagues including TPCB student Sophia Christodolou-Rubalcava in the labs of Prof. Yael David (MSK) and Prof. Shixin Liu (Rockefeller). Using a combination of protein chemistry and single-molecule biophysical techniques, the team demonstrated that linker histone H1 preferentially interacts with single-stranded DNA to promote phase separation, a process linked to the formation of many types of membraneless sub-compartments in the cell. In particular, they found that this phase separation occurs at single-stranded DNA in replication forks, structures that are formed when the genome is being copied. Replication forks become vulnerable if the DNA is damaged, leading to genome instability. Thus, this work suggests that H1 plays a role beyond simply compacting DNA and, by compartmentalizing the genome, may help maintain its stability when the cell is under stress. [Read more at: Nat. Struct. Mol. Biol.]
Each cell in the body contains some two meters of DNA. To compact this genetic material into the cell nucleus, a group of five proteins called histones collaborate to organize the DNA into a condensed yet readable format. Histone H1, also known as linker histone, has for many years been known to act in concert with the other four histone proteins to compact double-stranded genomic DNA. Together, these histone proteins pack away the vast majority of the genome that eventually form chromosomes as we know them. However, in a new report in Nature Structural and Molecular Biology, a collaborative TPCB team has discovered an unexpected and independent function for linker histone H1. The work was led by TPCB students Dr. Rachel Leicher, Dr. Adewola Osunsade, and Gabriella Chua, and postdoctoral fellow Dr. Sarah Faulkner, along with other colleagues including TPCB student Sophia Christodolou-Rubalcava in the labs of Prof. Yael David (MSK) and Prof. Shixin Liu (Rockefeller). Using a combination of protein chemistry and single-molecule biophysical techniques, the team demonstrated that linker histone H1 preferentially interacts with single-stranded DNA to promote phase separation, a process linked to the formation of many types of membraneless sub-compartments in the cell. In particular, they found that this phase separation occurs at single-stranded DNA in replication forks, structures that are formed when the genome is being copied. Replication forks become vulnerable if the DNA is damaged, leading to genome instability. Thus, this work suggests that H1 plays a role beyond simply compacting DNA and, by compartmentalizing the genome, may help maintain its stability when the cell is under stress. [Read more at: Nat. Struct. Mol. Biol.]
February 2022
SEAK and Destroy: Engineered CAR-T Cells Generate Small-Molecule Drugs in situ at Tumors
 CAR-T cells (chimeric antigen receptor T cells) are revolutionary cellular therapies currently being used to treat B-cell cancers. CAR-T cells are produced from patient’s own T cells, which are engineered to express a non-natural receptor that recognizes a tumor antigen and activates an antitumor immune response. However, despite this breakthrough, CAR-T cell therapies face several limitations, including immunosupression in the tumor microenvironment, limited penetration of solid tumors, and eventual relapse resulting from antigen-negative tumor cells. In a recent publication in Nature Chemical Biology, TPCB student J. Peter Lee, postdoctoral fellow Dr. Thomas Gardner, Prof. Derek Tan, Prof. David Scheinberg, and their coworkers at MSK have created a new class of CAR-T cells that have the added capability to generate small-molecule anticancer drugs in situ, which may synergize with the antitumor activity of the cells themselves, potentially overcoming several of the limitations of conventional CAR-T cells. These novel SEAKER (Synthetic Enzyme-Armed KillER) cells proliferate at tumors and express an activating enzyme that converts a non-toxic prodrug, which is administered systemically, to the active parent drug locally at the tumor site. The team demonstrated the effectiveness of this modular platform across several different enzyme–prodrug combinations, both in cell culture and in mouse tumor models. This work sets the stage for further investigations of this new therapeutic strategy for cancer and potentially other diseases. [Read more at: MSK News | Nat. Chem. Bio. | Nat. Rev. Drug Discov.. | Cancer Discov. | The Science Advisory Board | BioIT World | Redaccíon Médica (Spanish) | Read the article in: Nat. Chem. Biol. ]
CAR-T cells (chimeric antigen receptor T cells) are revolutionary cellular therapies currently being used to treat B-cell cancers. CAR-T cells are produced from patient’s own T cells, which are engineered to express a non-natural receptor that recognizes a tumor antigen and activates an antitumor immune response. However, despite this breakthrough, CAR-T cell therapies face several limitations, including immunosupression in the tumor microenvironment, limited penetration of solid tumors, and eventual relapse resulting from antigen-negative tumor cells. In a recent publication in Nature Chemical Biology, TPCB student J. Peter Lee, postdoctoral fellow Dr. Thomas Gardner, Prof. Derek Tan, Prof. David Scheinberg, and their coworkers at MSK have created a new class of CAR-T cells that have the added capability to generate small-molecule anticancer drugs in situ, which may synergize with the antitumor activity of the cells themselves, potentially overcoming several of the limitations of conventional CAR-T cells. These novel SEAKER (Synthetic Enzyme-Armed KillER) cells proliferate at tumors and express an activating enzyme that converts a non-toxic prodrug, which is administered systemically, to the active parent drug locally at the tumor site. The team demonstrated the effectiveness of this modular platform across several different enzyme–prodrug combinations, both in cell culture and in mouse tumor models. This work sets the stage for further investigations of this new therapeutic strategy for cancer and potentially other diseases. [Read more at: MSK News | Nat. Chem. Bio. | Nat. Rev. Drug Discov.. | Cancer Discov. | The Science Advisory Board | BioIT World | Redaccíon Médica (Spanish) | Read the article in: Nat. Chem. Biol. ]
January 2022
TPCB Faculty Honored with Major Awards and Grants
TPCB faculty have been honored recently with a number of prestigious awards and grants, recognizing their excellence in scientific research.
 • Prof. Seth Darst has been honored with the Gregori Aminoff Prize by The Royal Swedish Academy of Sciences. The award recognizes his work at Rockefeller to elucidate the structure of bacterial RNA polymerase and its interactions with tuberculosis antibiotics. He shares the prize with Elena Conti of the Max Planck Institute of Biochemistry and Patrick Cramer of the Max Planck Institute for Biophysical Chemistry. [Read more at: Royal Swedish Academy of Sciences | Rockefeller News]
• Prof. Seth Darst has been honored with the Gregori Aminoff Prize by The Royal Swedish Academy of Sciences. The award recognizes his work at Rockefeller to elucidate the structure of bacterial RNA polymerase and its interactions with tuberculosis antibiotics. He shares the prize with Elena Conti of the Max Planck Institute of Biochemistry and Patrick Cramer of the Max Planck Institute for Biophysical Chemistry. [Read more at: Royal Swedish Academy of Sciences | Rockefeller News]
 • Prof. Daniel Heller has been elected a Fellow of the American Institute for Medical and Biomedical Engineering. He was inducted in recognition of his outstanding contributions to nanotechnology for drug delivery and biosensors at MSK. The College of Fellows is comprised of top medical and biological engineers in the country, and are elected by their peers for outstanding contributions to engineering and medicine research, including pioneering new technologies. [Read more at: AIMBE | MSK News]
• Prof. Daniel Heller has been elected a Fellow of the American Institute for Medical and Biomedical Engineering. He was inducted in recognition of his outstanding contributions to nanotechnology for drug delivery and biosensors at MSK. The College of Fellows is comprised of top medical and biological engineers in the country, and are elected by their peers for outstanding contributions to engineering and medicine research, including pioneering new technologies. [Read more at: AIMBE | MSK News]
 • Prof. Kayvan Keshari has been recognized as a Distinguished Investigator by the Academy of Radiology & Biomedical Imaging Research. He is honored for his work using hyperpolarized magnetic resonance imaging probes to monitor cancer metabolism in real-time in animals and in patients. The award recognizes outstanding contributions in medical imaging. [Read more at: Academy for Radiology & Biomedical Imaging Research | MSK News]
• Prof. Kayvan Keshari has been recognized as a Distinguished Investigator by the Academy of Radiology & Biomedical Imaging Research. He is honored for his work using hyperpolarized magnetic resonance imaging probes to monitor cancer metabolism in real-time in animals and in patients. The award recognizes outstanding contributions in medical imaging. [Read more at: Academy for Radiology & Biomedical Imaging Research | MSK News]
 • Prof. Viviana Risca has been named a 2021 Rita Allen Foundation Scholar. This prestigious early-career award will support her work at Rockefeller to understand the molecular mechanism by which a protein called macroH2A regulates DNA folding in cells and suppresses cancer. She joins five other TPCB faculty who have received this honor during the early stages of their careers. [Read more at: Rita Allen Foundation]
• Prof. Viviana Risca has been named a 2021 Rita Allen Foundation Scholar. This prestigious early-career award will support her work at Rockefeller to understand the molecular mechanism by which a protein called macroH2A regulates DNA folding in cells and suppresses cancer. She joins five other TPCB faculty who have received this honor during the early stages of their careers. [Read more at: Rita Allen Foundation]
 • Prof. David Scheinberg has received an NIH R35 Outstanding Investigator Award from the National Cancer Institute. The 7-year grant will support his innovative research program at MSK developing antibody-based and cellular therapeutics. This award supports accomplished leaders in cancer research who are contributing to the understanding of cancer and breakthrough applications of that knowledge. [Read more at: NCI | MSK News]
• Prof. David Scheinberg has received an NIH R35 Outstanding Investigator Award from the National Cancer Institute. The 7-year grant will support his innovative research program at MSK developing antibody-based and cellular therapeutics. This award supports accomplished leaders in cancer research who are contributing to the understanding of cancer and breakthrough applications of that knowledge. [Read more at: NCI | MSK News]
 • Prof. Ekaterina Vinogradova has been named a Searle Scholar and a Damon Runyon–Rachleff Innovation awardee. These early career awards will support her research at Rockefeller using chemoproteomic approaches to study cytokine release storm and the mechanisms of T-cell exhaustion in cancer, respectively. She is the fifth TPCB faculty member to be named a Searle Scholar and the first to receive the Damon Runyon Innovation award. [Read more at: Searle Scholars Program | Damon Runyon Foundation]
• Prof. Ekaterina Vinogradova has been named a Searle Scholar and a Damon Runyon–Rachleff Innovation awardee. These early career awards will support her research at Rockefeller using chemoproteomic approaches to study cytokine release storm and the mechanisms of T-cell exhaustion in cancer, respectively. She is the fifth TPCB faculty member to be named a Searle Scholar and the first to receive the Damon Runyon Innovation award. [Read more at: Searle Scholars Program | Damon Runyon Foundation]
November 2021
Vanessa Ruta Named HHMI Investigator
 TPCB faculty member Prof. Vanessa Ruta has been named an Investigator of the Howard Hughes Medical Institute. She is one of 33 new HHMI Investigators selected this year, each of whom will receive approximately $9M in research funding over a 7-year term. Prof. Ruta’s lab at Rockefeller studies the molecular and neural mechanisms of olfaction in Drosophila melanogaster, exploring both the structural basis of odor detection at the receptor level and neural algorithms of odor navigation at the behavioral level. She joins six other current HHMI Investigators on the TPCB faculty. [Read more at: HHMI | Rockefeller News]
TPCB faculty member Prof. Vanessa Ruta has been named an Investigator of the Howard Hughes Medical Institute. She is one of 33 new HHMI Investigators selected this year, each of whom will receive approximately $9M in research funding over a 7-year term. Prof. Ruta’s lab at Rockefeller studies the molecular and neural mechanisms of olfaction in Drosophila melanogaster, exploring both the structural basis of odor detection at the receptor level and neural algorithms of odor navigation at the behavioral level. She joins six other current HHMI Investigators on the TPCB faculty. [Read more at: HHMI | Rockefeller News]
October 2021
Under Construction: New Molecular Insights into Protein Synthesis by the Ribosome
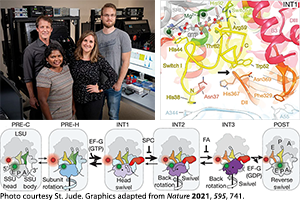 Ribosomes are miniature factories within the cell that produce proteins. The molecular mechanisms of ribosomal protein synthesis have been studied for decades, but numerous question remain regarding how this complex process is orchestrated and driven energetically. Now, TPCB student Emily Rundlet and Prof. Scott Blanchard, formerly of Weill Cornell and currently on the faculty at St. Jude Children’s Hospital, and coworkers have used single-molecule fluorescence imaging and cryoelectron microscopy to provide important new insights into this process. Their work, which was published recently in Nature, focuses on translocation, whereby the ribosome orchestrates the movement of aminoacyl-tRNA building blocks and mRNA templates through its active site to synthesize a specific protein sequence according to the genetic code. They first used single-molecule FRET (Förster resonance energy transfer) experiments to identify conditions and small-molecule antibiotics that could trap the ribosome in intermediate states for structural characterization. This led to the surprising discovery that EF-G (elongation factor G), a GTP-dependent enzyme that drives translocation, engages the ribosome complex at an early stage in translocation with its GTP intact. This indicated that the chemical energy released by GTP hydrolysis is not used until later in the translocation process. The lab’s ongoing efforts are now focused on elucidating the molecular details of those downstream steps. These studies provide a deeper understanding of this fundamental cellular process and may shed light on ribosome dysfunction in disease states, as well as set the stage for the development of new antibiotics that disrupt ribosome function. [Read more at: St. Jude News (Lay Summary) | Read the article in: Nature ]
Ribosomes are miniature factories within the cell that produce proteins. The molecular mechanisms of ribosomal protein synthesis have been studied for decades, but numerous question remain regarding how this complex process is orchestrated and driven energetically. Now, TPCB student Emily Rundlet and Prof. Scott Blanchard, formerly of Weill Cornell and currently on the faculty at St. Jude Children’s Hospital, and coworkers have used single-molecule fluorescence imaging and cryoelectron microscopy to provide important new insights into this process. Their work, which was published recently in Nature, focuses on translocation, whereby the ribosome orchestrates the movement of aminoacyl-tRNA building blocks and mRNA templates through its active site to synthesize a specific protein sequence according to the genetic code. They first used single-molecule FRET (Förster resonance energy transfer) experiments to identify conditions and small-molecule antibiotics that could trap the ribosome in intermediate states for structural characterization. This led to the surprising discovery that EF-G (elongation factor G), a GTP-dependent enzyme that drives translocation, engages the ribosome complex at an early stage in translocation with its GTP intact. This indicated that the chemical energy released by GTP hydrolysis is not used until later in the translocation process. The lab’s ongoing efforts are now focused on elucidating the molecular details of those downstream steps. These studies provide a deeper understanding of this fundamental cellular process and may shed light on ribosome dysfunction in disease states, as well as set the stage for the development of new antibiotics that disrupt ribosome function. [Read more at: St. Jude News (Lay Summary) | Read the article in: Nature ]
September 2021
TPCB Hosts First Hybrid Tri-Institutional Chemical Biology Symposium
TPCB presented the 17th Annual Tri-Institutional Chemical Biology Symposium on August 31, 2021. In a preview of how many events are likely to be conducted in the coming academic year, the symposium was convened in a hybrid format, with TPCB students and faculty attending in person while other guests tuned in remotely via Zoom webinar. All in-person attendees were fully vaccinated with talks presented in a large auditorium at under 20% of its normal capacity (although institutional protocols currently permit 75% occupancy), and universal masking, including speakers. Since the beginning of the COVID-19 pandemic, TPCB has been at the forefront of leveraging new meeting formats to share scientific information and to promote interactions within the community. This year’s event was attended by some 250 registrants from around the world and included faculty keynote presentations, student seminars, and an online poster session featuring nearly 50 presentations representing 10 different institutions on four continents.


[First photo]: TPCB student co-organizer Lauren Vostal opens the first hybrid Tri-Institutional Chemical Biology Symposium. [Second photo]: Symposium speakers and organizers (from top left): Prof. Kate Carroll, Natalie Jones, TPCB Director Prof. Derek Tan, Hsin-che Huang, J. Jeya Vandana, Lauren Vostal, Prof. Bil Clemons, Igor Maksimovic, Emily Rundlet, Prof. Shuibing Chen
Prof. Kate Carroll (below) of Scripps kicked off the event with a virtual presentation on her lab’s investigations of redox signaling in metazoans via cysteine oxidation by hydrogen peroxide. She described chemical probes developed by her group to detect specific cysteine oxidation states across the proteome, such as electrophilic nitrogen probes for sulfinic acids and phosphine ylides for sulfenic acids. Next, TPCB student Natalie Jones (below), presenting remotely from California, shared her work with Prof. Tarun Kapoor at Rockefeller, where she has developed a bump–hole approach to provide selective, covalent chemical probes of AAA proteins involved in cytoskeletal rearrangements via introduction of an engineered cysteine residue.


Hybrid seminar presentations by Prof. Kate Carroll (first photo) and TPCB student Natalie Jones (second photo).
In the first in-person presentation, TPCB student Igor Maksimovic (below) from Prof. Yael David’s lab at MSK described his work developing chemical tools to study a non-enzymatic chromatin modification called glycation, with a particular focus on the functions of the deglycation enzyme FN3K. He also presented related efforts to develop small-molecule inhibitors of another deglycase DJ-1 that is implicated in cancer. In the final talk of the morning, Prof. Bil Clemons (below) of Caltech began by sharing his profound perspectives of the 1619 Project and the history of slavery and racism in America. He then presented his lab’s work elucidating the structure basis for bacterial lysis by the bacteriophage single gene lysis antibiotic ‘protein E’, via recruitment of SlyD peptidyl prolyl isomerase to the essential bacterial translocase MraY.


TPCB student Igor Maksimovic (first photo) and Prof. Bil Clemons (second photo) close out the morning session of the symposium.
The in-person attendees then enjoyed an outdoor boxed lunch at the Belfer Terrace on the Weill Cornell Campus (below), which was followed by a virtual poster session featuring presentations by 45 graduate students, postdoctoral fellows, and faculty from around the world. The organizers leveraged experience from TPCB’s successful poster session at last January’s Interview & Recruiting Open House to stage a dynamic and engaging event using Zoom breakout rooms. Poster presentations were judged by the keynote speakers and TPCB faculty members, with awards sponsored by TPCB and our promotional partners.

Keynote speakers and TPCB students enjoy lunch outdoors at the Belfer Terrace on the Weill Cornell campus.
TPCB Graduate Students
• Nathan Harper, TPCB (Klinge Lab, Rockefeller)
– Late Maturation Events of the Human Mitochondrial Small Subunit Captured by Cryo-EM
– Chemical Science Prize
• Jakob Hebert, TPCB (David Lab, MSK)
– The SWI/SNF Related Protein SMARCA3 is a Histone H3K23 Ubiquitin Ligase that Regulates H3K9me3 in Cancer
– Nature Chemical Biology and ChemBioChem Prizes
• Hsin-Che Huang, TPCB (Bachovchin Lab, MSK)
– Characterizing the Role of the INO80 Complex in DPP8/9 Inhibition-Induced Pyroptosis
– RSC Chemical Biology Prize
• Ilana Kotliar, TPCB (Sakmar Lab, Rockefeller)
– Multiplexed Analysis of the Secretin-like GPCR-RAMP Interactome by Suspension Bead Array
– RSC Chemical Biology Prize
• Jordan Mattheisen, TPCB (Sakmar Lab, Rockefeller)
– Targeting Cryptic Drug-Binding Pockets in GPCRs using Genetic Code Expansion Technology
– Organic & Biomolecular Chemistry Prize
Open Graduate Students
• Anna Impastato, New York University (Dirk Trauner Lab)
– Optical Control of Mitosis with a Photoswitchable Eg5 Inhibitor
– Nature Chemical Biology and ChemBioChem Prizes
• Kleo Palate, University of York (Will Unsworth Lab)
– Expanding the Scope of Successive Ring Expansion
– Cell Chemical Biology Prize
Senior Scientists
• Prof. David Lapinsky, Duquesne University School of Pharmacy
– Appendage- and Scaffold-Diverse Electrophilic and Photoreactive Fully Functionalized Small-Molecule Probes for Integrated Phenotypic Screening-Target Identification Campaigns Via Minimalist Trifunctional Building Blocks
– Nature Chemical Biology and ChemBioChem Prizes
• Dr. Johnson Luwang, Indian Institute of Science Education and Research, Thiruvananthapuram (Ramanathan Natesh Lab)
– Impingement of the p53 Stability by Virtue of Tetramerization
– Cell Chemical Biology Prize
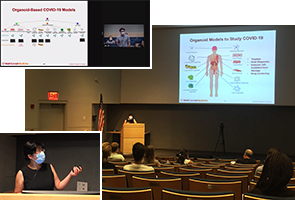

TPCB faculty member Prof. Shuibing Chen of Weill Cornell (first photo) and student Emily Rundlet (second photo) open the afternoon seminar session.
The afternoon talks kicked off with a seminar by Prof. Shuibing Chen (above) of Weill Cornell and TPCB, who presented her recent work in developing organoid models to study viral pathogens and to search for new antivirals to treat COVID-19. In particular, evaluation of SARS-CoV-2 infection of organoids models from different tissues revealed high infectivity in pancreatic beta cells, leading to the discovery of transdifferentation of these cells in vitro and in patient samples. This model was also used to identify small molecules that inhibit macrophage-mediated ROS production in response to SARS-CoV-2 infection of cardiomyocyte cells, providing a potential means to block virus-induced damage. Next, TPCB student Emily Rundlet (above) returned to NYC to speak about her work with Prof. Scott Blanchard, formerly at Weill Cornell and currently at St. Jude’s Children’s Research Hospital. In her recently published work, she used single-molecule FRET and cryo-EM approaches to elucidate the dynamic movement of elongation factor EF-G in guiding tRNA translocation across the ribosome active site.


[First photo]: Prof. Stuart Schreiber presents the final faculty keynote address of the symposium. [Second photo]: TPCB students hosted the faculty keynote speakers for an outdoor dinner at a favorite local restaurant.
The scientific events concluded with a remote presentation by Prof. Stuart Schreiber (above) of The Broad Institute and Harvard, in which he gave a historical overview of the development of molecular glues over the last three decades, starting with natural products such as FK506, cyclosporin A, and rapamycin, and leading to more recent advances in developing molecular glues that induce protein degradation as novel therapeutics. Following the poster award presentations, the TPCB students and keynote speakers enjoyed an outdoor dinner together at a local favorite restaurant, Bottega.
This first hybrid sympsosium was made possible by the efforts of TPCB student organizers Hsin-Che Huang, Lauren Vostal, and Jeye Vandana, who charted new waters in navigating this new format. The organizers were assisted by TPCB faculty mentor Prof. Daniel Heller of MSK, TPCB Program Coordinator Margie Hinonangan-Mendoza, and TPCB Director Prof. Derek Tan. The event was simulcast with support from the MSK Department of Marketing & Communications, in particular Jennifer Brosnan, Senior Project Manager and Sam Palmucci, Video Department Manager. Promotional support was provided by ACS Chemical Biology, Biochemistry, Cell Chemical Biology, ChemBioChem, Chemical Science, Nature Chemical Biology, Organic & Biomolecular Chemistry, RSC Chemical Biology, and the New York Academy of Sciences.
Nicholas Prescott Awarded Prestigious Fellowship from the National Cancer Institute
 In 2016, the National Cancer Institute (NCI) established a new award for graduate students to facilitate their transition to successful postdoctoral appointments and to support their ongoing career development toward becoming independent investigators. TPCB student Nicholas Prescott was one of 24 students nationwide to receive this prestigious NCI Predoctoral to Postdoctoral Fellow Transition Award (F99/K00) this year! Nick is currently a 5th year student working with Prof. Yael David at MSK. His research focuses on understanding how DNA viruses such as hepatitis B interact with chromatin factors in human host cells, which can contribute to the development of cancer. During his graduate training, he has developed a platform to assemble the hepatitis B chromosome in vitro and study its interplay with host and viral proteins. After he completes his PhD, he plans to continue onto postdoctoral training in which he will investigate chromatin remodeling enzymes and their molecular role in carcinogenesis. The F99/K00 award will provide up to 2 years of funding in the predoctoral phase, followed by up to 4 years of funding in the postdoctoral phase. [Read more at: Weill Cornell News]
In 2016, the National Cancer Institute (NCI) established a new award for graduate students to facilitate their transition to successful postdoctoral appointments and to support their ongoing career development toward becoming independent investigators. TPCB student Nicholas Prescott was one of 24 students nationwide to receive this prestigious NCI Predoctoral to Postdoctoral Fellow Transition Award (F99/K00) this year! Nick is currently a 5th year student working with Prof. Yael David at MSK. His research focuses on understanding how DNA viruses such as hepatitis B interact with chromatin factors in human host cells, which can contribute to the development of cancer. During his graduate training, he has developed a platform to assemble the hepatitis B chromosome in vitro and study its interplay with host and viral proteins. After he completes his PhD, he plans to continue onto postdoctoral training in which he will investigate chromatin remodeling enzymes and their molecular role in carcinogenesis. The F99/K00 award will provide up to 2 years of funding in the predoctoral phase, followed by up to 4 years of funding in the postdoctoral phase. [Read more at: Weill Cornell News]
Melinda Diver, Jacob Geri, and Jeremy Rock join the TPCB Faculty
TPCB is pleased to welcome three new faculty members to the program. They bring new research interests in the structural and mechanistic basis for pain sensation, in optically controlled proximity labeling technologies to probe protein interactions, and in chemical genomics studies of host-pathogen interactions in tuberculosis.
 • Prof. Melinda Diver joined the Memorial Sloan Kettering faculty in the Structural Biology Program in September 2021. Her lab seeks to understand at a molecular level how the body senses pain, in particular through the use of ion channels in neurons. They will use a comprehensive approach comprising high-resolution structure determination, electrophysiology, and chemical probes to study the normal physiology of pain sensation as well as how defects in pain sensation can lead to persistent pain disorders. Prof. Diver completed her PhD studies in biochemistry and structural biology with Prof. Stephen Long at MSK and Weill Cornell in 2014 and continued onto postdoctoral training investigating the cold and menthol-sensing channel TRPM8 with Prof. David Julius at UCSF. [Read more]
• Prof. Melinda Diver joined the Memorial Sloan Kettering faculty in the Structural Biology Program in September 2021. Her lab seeks to understand at a molecular level how the body senses pain, in particular through the use of ion channels in neurons. They will use a comprehensive approach comprising high-resolution structure determination, electrophysiology, and chemical probes to study the normal physiology of pain sensation as well as how defects in pain sensation can lead to persistent pain disorders. Prof. Diver completed her PhD studies in biochemistry and structural biology with Prof. Stephen Long at MSK and Weill Cornell in 2014 and continued onto postdoctoral training investigating the cold and menthol-sensing channel TRPM8 with Prof. David Julius at UCSF. [Read more]
 • Prof. Jacob Geri launched his lab in the Pharmacology Department at Weill Cornell Medicine in September 2021. His lab is developing optically-controlled proximity labeling technologies to identify protein interactions that drive disease phenotypes with high spatial and temporal resolution. His multidisciplinary collaborative work includes efforts in photochemistry, organic and inorganic synthesis, and molecular biology. Prof. Geri completed his PhD degree in chemistry with Prof. Nathaniel Szymczak at the University of Michigan in 2018, and postdoctoral studies in photocatalytic chemistry with Prof. David MacMillan at Princeton University. [Read more]
• Prof. Jacob Geri launched his lab in the Pharmacology Department at Weill Cornell Medicine in September 2021. His lab is developing optically-controlled proximity labeling technologies to identify protein interactions that drive disease phenotypes with high spatial and temporal resolution. His multidisciplinary collaborative work includes efforts in photochemistry, organic and inorganic synthesis, and molecular biology. Prof. Geri completed his PhD degree in chemistry with Prof. Nathaniel Szymczak at the University of Michigan in 2018, and postdoctoral studies in photocatalytic chemistry with Prof. David MacMillan at Princeton University. [Read more]
 • Prof. Jeremy Rock joined TPCB in January 2021 and is a faculty member at The Rockefeller University. His lab uses functional and chemical genomic approaches to investigate host–pathogen interactions in tuberculosis. These efforts include the identification of new antibiotic drug targets and small-molecule inhibitors with novel mechanisms of action. Prof. Rock completed his PhD training in genetics with Prof. Angelika Amon at MIT, then continued onto postdoctoral studies with Prof. Sarah Fortune and Prof. Eric Rubin at Harvard, where he developed a CRISPR platform for gene interference in Mycobacterium tuberculosis. [Read more]
• Prof. Jeremy Rock joined TPCB in January 2021 and is a faculty member at The Rockefeller University. His lab uses functional and chemical genomic approaches to investigate host–pathogen interactions in tuberculosis. These efforts include the identification of new antibiotic drug targets and small-molecule inhibitors with novel mechanisms of action. Prof. Rock completed his PhD training in genetics with Prof. Angelika Amon at MIT, then continued onto postdoctoral studies with Prof. Sarah Fortune and Prof. Eric Rubin at Harvard, where he developed a CRISPR platform for gene interference in Mycobacterium tuberculosis. [Read more]
August 2021
ChBSP Summer Internships Return to Campus
The Tri-Institutional Chemical Biology Summer Program (ChBSP) was pleased to welcome this year’s class of summer interns on campus for in-person research experiences! Due to COVID-19-related campus access restrictions, all of the interns were hosted in MSK labs, with faculty and TPCB students from Rockefeller and Weill Cornell labs hosting other academic events for the interns. Activities included a journal club mentored by TPCB students Emma Garst and Natalie Jones, seminars presented by TPCB faculty members, and career development workshops. At the end of the summer, the ChBSP students presented their research to the entire Tri-Institutional community at an online poster session on August 12, 2021, demonstrating their impressive accomplishments in chemical biology over the summer.
 Dominic Covelli
Dominic Covelli
Brown University, Class of 2022
Molecular Mechanisms Underlying Inflammasome Formation
with Prof. Daniel Bachovchin, Sloan Kettering
 Julia Flood
Julia Flood
Fordham University, Class of 2022
Epigenetic Regulation of Transcription
with Prof. Yael David, Sloan Kettering
 Amanda Garza
Amanda Garza
Boston College, Class of 2022
Imidazopyrazine-based Autophagy Modulators for Alzheimer’s Disease
with Prof. Yueming Li, Sloan Kettering
 Maya Huffman
Maya Huffman
Williams College, Class of 2022
Direct Conversion of Carboxylic Acids to 3-Oxetanol Bioisosteres
with Prof. Derek Tan, Sloan Kettering
 Atara Neugroschl
Atara Neugroschl
Yeshiva University, Class of 2022
A Ubiquitin-independent Proteasome Pathway Activates the CARD8 Inflammasome
with Prof. Daniel Bachovchin, Sloan Kettering
 Abigail Pierre
Abigail Pierre
Seton Hall University, Class of 2022
PROTAC Degradation of Ribosomal Proteins
with Prof. Heeseon An, Sloan Kettering
The program was one of the few internships in the country that was able to proceed in person, thanks to the outstanding efforts of the program leadership, TPCB faculty member Prof. Daniel Bachovchin and Dr. Ushma Neill, Vice President of Scientific Education & Training at MSK, with generous financial support from the Sanders Innovation and Education Initiative of the Tri-Institutional Therapeutics Discovery Institute, led by Dr. Peter Meinke. [Read more]
Katya Vinogradova Named to C&EN Talented 12
 Each year since 2015, Chemical & Engineering News has recognized 12 talented young scientists who are taking risks in the early stages of their careers to tackle global problems. Congratulations to new TPCB faculty member Prof. Katya Vinogradova who has been selected to this year’s class! Prof. Vinogradova’s lab at Rockefeller University combines organic synthesis and chemical proteomics to study proteins involved in the human immune system. In particular, the lab works to discover chemical probes that selectively label specific amino acid residues on proteins, thereby perturbing their activity, interactions, and expression. They seek to use these probes to study the molecular pathways involved in immune regulation, including in autoimmune disorders. Prof. Vinogradova carried out her undergraduate and master’s studies at the Higher Chemical College of the Russian Academy of Sciences (a department of Mendeleev University of Chemical Technology). She completed her PhD in organometallic chemistry with Prof. Steven Buchwald at MIT, then postdoctoral training in chemical proteomics with Prof. Benjamin Cravatt at the Scripps Research Institute. She joined Rockefeller University and the TPCB faculty in late 2020. [Read more at: C&EN]
Each year since 2015, Chemical & Engineering News has recognized 12 talented young scientists who are taking risks in the early stages of their careers to tackle global problems. Congratulations to new TPCB faculty member Prof. Katya Vinogradova who has been selected to this year’s class! Prof. Vinogradova’s lab at Rockefeller University combines organic synthesis and chemical proteomics to study proteins involved in the human immune system. In particular, the lab works to discover chemical probes that selectively label specific amino acid residues on proteins, thereby perturbing their activity, interactions, and expression. They seek to use these probes to study the molecular pathways involved in immune regulation, including in autoimmune disorders. Prof. Vinogradova carried out her undergraduate and master’s studies at the Higher Chemical College of the Russian Academy of Sciences (a department of Mendeleev University of Chemical Technology). She completed her PhD in organometallic chemistry with Prof. Steven Buchwald at MIT, then postdoctoral training in chemical proteomics with Prof. Benjamin Cravatt at the Scripps Research Institute. She joined Rockefeller University and the TPCB faculty in late 2020. [Read more at: C&EN]
Congratulations to TPCB’s 2021 Graduates
TPCB congratulates our banner group of 2021 graduates, Didar Ciftci (Boudker Lab, WCM), Emma Garst (Hang Lab, RU), Mehtap Isik (Chodera Lab, MSK), Peter Lee (Tan Lab, MSK), Rachel Leicher (Liu Lab, RU), Fangyu Liu (Jue Chen Lab, RU), Lin Mei (Alushin Lab, RU), Lina Miller (Klinge Lab, RU), Wola Osunsade (David Lab, MSK), Sahana Rao (Bachovchin Lab, MSK), Zheng Ser (Kentsis Lab, MSK), Taku Tsukidate (Hang Lab, RU), and Rafal Wiewiora (Chodera Lab, MSK)!
These exceptional students successfully completed their PhD training in the midst of the COVID-19 pandemic, between June 2020 and July 2021. They demonstrated outstanding dedication and resilience, continuing their work in the laboratories, publishing their work in leading scientific journals, and presenting their work at virtual thesis defenses, surrounded remotely by colleagues, friends, and family. Many of these students received their PhD degrees at the Weill Cornell Commencement Ceremony on May 21, 2021 and at The Rockefeller University Convocation Ceremony on June 10, 2021. Students in MSK labs were also recognized at the MSK Academic Convocation on May 19, 2021. The remaining graduates will be recognized formally at next year’s ceremonies. We wish our newest alumni all the best as they continue onto the next stages of their careers! [Read more at: Weill Cornell | Rockefeller (Video) | Memorial Sloan Kettering]
 • Hatice Didar Ciftci, PhD – “Linking Dynamics and Function in Glutamate Transporters at Single Molecule Resolution”
• Hatice Didar Ciftci, PhD – “Linking Dynamics and Function in Glutamate Transporters at Single Molecule Resolution”
TPCB Mentor: Prof. Olga Boudker, Weill Cornell Medicine
Current Position: Postdoctoral Fellow, Prof. Xiaowei Zhuang, Harvard University
 • Emma Garst, PhD – “Reconstitution and Biophysical Analysis of Site-specifically Lipidated IFITM3”
• Emma Garst, PhD – “Reconstitution and Biophysical Analysis of Site-specifically Lipidated IFITM3”
TPCB Mentor: Prof. Howard Hang, The Rockefeller University
Current Position: Postdoctoral Fellow, Prof. James Hurley, University of California, Berkeley
 • Mehtap Isik, PhD – “Advancing Small Molecule Physicochemical Property Predictions for Computational Drug Discovery”
• Mehtap Isik, PhD – “Advancing Small Molecule Physicochemical Property Predictions for Computational Drug Discovery”
TPCB Mentor: Prof. John Chodera, Memorial Sloan Kettering
Current Position: Computational Chemistry & Cheminformatics Scientist, Moderna
 • Jonghan Peter Lee, PhD – “Targetable Cellular Micropharmacies: Engineering CAR T Cells to Activate Small Molecule Prodrugs”
• Jonghan Peter Lee, PhD – “Targetable Cellular Micropharmacies: Engineering CAR T Cells to Activate Small Molecule Prodrugs”
TPCB Mentor: Prof. Derek Tan, Memorial Sloan Kettering
Current Position: Senior Consultant, Trinity Life Sciences
 • Rachel Leicher, PhD – “Single-molecule Investigation of Chromatin-associated Factors in Genome Organization and Epigenetic Maintenance”
• Rachel Leicher, PhD – “Single-molecule Investigation of Chromatin-associated Factors in Genome Organization and Epigenetic Maintenance”
TPCB Mentor: Prof. Shixin Liu, The Rockefeller University
Current Position: Scientist, Constellation Pharmaceuticals
 • Fangyu Liu, PhD – “Structural Study of Disease Relevant ABC Transporters-Cystic Fibrosis Transmembrane Conductance Regulator and ABCA4”
• Fangyu Liu, PhD – “Structural Study of Disease Relevant ABC Transporters-Cystic Fibrosis Transmembrane Conductance Regulator and ABCA4”
TPCB Mentor: Prof. Jue Chen, The Rockefeller University
Current Position: Postdoctoral Fellow, Prof. Brian Shoichet, UCSF
 • Lin Mei, PhD – “Multi-modal Regulation of Actin Networks”
• Lin Mei, PhD – “Multi-modal Regulation of Actin Networks”
TPCB Mentor: Prof. Gregory Alushin, The Rockefeller University
Current Position: Postdoctoral Fellow, Prof. John Rubinstein, Hospital for Sick Children, University of Toronto
 • Linamarie Miller, PhD – “Structural Studies of the Nucleolar Stages of Ribosome Biogenesis in Yeast”
• Linamarie Miller, PhD – “Structural Studies of the Nucleolar Stages of Ribosome Biogenesis in Yeast”
TPCB Mentor: Prof. Sebastian Klinge, The Rockefeller University
Current Position: Postdoctoral Fellow, Prof. James Shorter, University of Pennsylvania
 • Adewola Osunsade, PhD – “Biochemical and Biophysical Characterization of Human Linker Histone Variants”
• Adewola Osunsade, PhD – “Biochemical and Biophysical Characterization of Human Linker Histone Variants”
TPCB Mentor: Prof. Yael David, Memorial Sloan Kettering
Current Position: Consultant, McKinsey & Co.
 • Sahana Rao, PhD – “Discovery and Characterization of NLRP1 Inflammasome Activators”
• Sahana Rao, PhD – “Discovery and Characterization of NLRP1 Inflammasome Activators”
TPCB Mentor: Prof. Daniel Bachovchin, Memorial Sloan Kettering
Current Position: Postdoctoral Fellow, Prof. Vamsi Mootha, Massachusetts General Hospital
 • Zheng Ser, PhD – “Elucidation of Structure and Interactions of DNA Binding Protein Complexes Through Mass Spectrometry Proteomics”
• Zheng Ser, PhD – “Elucidation of Structure and Interactions of DNA Binding Protein Complexes Through Mass Spectrometry Proteomics”
TPCB Mentor: Prof. Alex Kentsis, Memorial Sloan Kettering
Current Position: Postdoctoral Fellow, Dr. Radoslaw Sobota, IMCB, A*STAR Singapore
 • Taku Tsukidate, PhD – “Chemical Tools for Exploring Metabolite Interactions with Nuclear Receptors and Beyond”
• Taku Tsukidate, PhD – “Chemical Tools for Exploring Metabolite Interactions with Nuclear Receptors and Beyond”
TPCB Mentor: Prof. Howard Hang, The Rockefeller University
Current Position: Postdoctoral Fellow, Dr. Shawn Xuanwen Li, Merck Research Labs
 • Rafal Wiewora, PhD – “Rigorous Construction of Markov State Models for Conformationally Selective Drug
• Rafal Wiewora, PhD – “Rigorous Construction of Markov State Models for Conformationally Selective Drug
Design”
TPCB Mentor: Prof. John Chodera, Memorial Sloan Kettering
Current Position: Senior Investigator, Silicon Therapeutics / Roivant Sciences







April 2021
Luciano Marraffini Elected to American Academy of Arts and Sciences
 TPCB faculty member Prof. Luciano Marraffini has been elected to the American Academy of Arts and Sciences. The Academy is an honorary society that was established in 1780 to honor the excellence of its members as well as to covene leaders from across disciplines to address significant challenges. Prof. Marraffini’s lab at Rockefeller University studies the molecular mechanisms of bacterial immunity to viruses using CRISPR-Cas systems. These systems have also served as the basis for gene editing approaches that are used widely in biomedical research and are being explored as treatments for diseases. Prof. Marraffini joins seven other TPCB faculty who have been recognized with this honor. [Read more at: AAAS]
TPCB faculty member Prof. Luciano Marraffini has been elected to the American Academy of Arts and Sciences. The Academy is an honorary society that was established in 1780 to honor the excellence of its members as well as to covene leaders from across disciplines to address significant challenges. Prof. Marraffini’s lab at Rockefeller University studies the molecular mechanisms of bacterial immunity to viruses using CRISPR-Cas systems. These systems have also served as the basis for gene editing approaches that are used widely in biomedical research and are being explored as treatments for diseases. Prof. Marraffini joins seven other TPCB faculty who have been recognized with this honor. [Read more at: AAAS]
October 2020
May the Force Be with You: How Cells Use Actin to Detect Mechanical Signals
 Cells respond to a wide range of chemical signals in their environment, using receptors that transmit signals from the cell surface to the nucleus, thus controlling cellular processes such as proliferation and death. In a new study published in the journal eLife, TPCB student Lin Mei and Prof. Gregory Alushin at Rockefeller, show how cells also detect mechanical signals from their environment using the multifunctional cytoskeletal protein actin, which forms filaments within cells. The team hypothesized that external mechanical tension might be transmitted through the cell by the actin filaments. Using single-molecule techniques, they demonstrated that a cell–cell adhesion protein, α-catenin, bound more tightly to actin filaments that were under mechanical tension. In contrast, another homologous adhesion protein, vinculin, did not respond to force on actin. Structural studies using cryoelectron microscopy pointed to a C-terminal tail found only in α-catenin as a potential source of this functional difference, and this was confirmed using a truncated mutant that showed no increase in binding in response to actin tension. This work sets the stage for further studies of the molecular and cellular mechanisms of mechanotransduction by cytoskeletal proteins and may one day open the door to the development of new therapeutics to treat diseases associated with defects in this signaling, including muscular dystrophies, cardiomyopathies, and metastatic cancer. TPCB student Rachel Leicher and Prof. Shixin Liu, both at Rockefeller, also contributed to the work. [Read more at: Rockefeller News | Read the article in: eLife]
Cells respond to a wide range of chemical signals in their environment, using receptors that transmit signals from the cell surface to the nucleus, thus controlling cellular processes such as proliferation and death. In a new study published in the journal eLife, TPCB student Lin Mei and Prof. Gregory Alushin at Rockefeller, show how cells also detect mechanical signals from their environment using the multifunctional cytoskeletal protein actin, which forms filaments within cells. The team hypothesized that external mechanical tension might be transmitted through the cell by the actin filaments. Using single-molecule techniques, they demonstrated that a cell–cell adhesion protein, α-catenin, bound more tightly to actin filaments that were under mechanical tension. In contrast, another homologous adhesion protein, vinculin, did not respond to force on actin. Structural studies using cryoelectron microscopy pointed to a C-terminal tail found only in α-catenin as a potential source of this functional difference, and this was confirmed using a truncated mutant that showed no increase in binding in response to actin tension. This work sets the stage for further studies of the molecular and cellular mechanisms of mechanotransduction by cytoskeletal proteins and may one day open the door to the development of new therapeutics to treat diseases associated with defects in this signaling, including muscular dystrophies, cardiomyopathies, and metastatic cancer. TPCB student Rachel Leicher and Prof. Shixin Liu, both at Rockefeller, also contributed to the work. [Read more at: Rockefeller News | Read the article in: eLife]
September 2020
TPCB Hosts 16th Annual Tri-Institutional Chemical Biology Symposium Online
TPCB sponsored the 16th Annual Tri-Institutional Chemical Biology Symposium on September 1, 2020. The event was held online for the first time due to the continuing COVID-19 pandemic, and the organizers leveraged this opportunity to expand registration dramatically to over 350 scientists from around the world. The program represented the breadth of the field and included faculty keynote lectures, student seminars, and a vibrant online poster session with some 50 presentations from 11 different institutions.

Symposium speakers and organizers (from top left): Prof. Lewis Cantley, Prof. Marcey Waters, Prof. Michael Fischbach, Prof. Livia Eberlin, Mizuho Horioka, Adewola Osunsade, Tandrila Das, Gabriella Chua, Ilana Kotliar, Lucas Repeta, and TPCB Director Prof. Derek Tan
Prof. Lewis Cantley (below) of Weill Cornell Medicine and TPCB opened the program with a presentation on the discovery and development of novel phosphoglycerate dehydrogenase inhibitors to treat cancer brain metastases. TPCB student Mizuho Horioka (below) then presented her studies of aberrant signaling and receptor bias in the G-protein-coupled receptor CYSLTR2, a driver oncogene in uveal melanoma, with Prof. Thomas Sakmar at Rockefeller. The next student presentation by TPCB student Adewola Osunsade (below) described her development of the first robust method to express and purify H1 linker histone variants and use of these proteins to study the impacts of H1 mutations on chromatin compaction and crosstalk with post-translational modifications on core histones, work carried out with Prof. Yael David at MSK. In the final talk of the morning, Prof. Marcey Waters (below) of UNC Chapel Hill shared her lab’s recent studies using genetic code expansion to investigate the role of cation-π interactions in recognition of trimethyl-lysine and crotonyl-lysine, and use of this information to develop selective reader proteins to identify histone marks.



Seminar presentations (from top left) by Prof. Lewis Cantley, Mizuho Horioka, Adewola Osunsade, and Prof. Marcey Waters
After a lunch break, the virtual poster session proved to be a highlight of the event, featuring poster presentations by over 50 graduate students, undergraduates, postbaccalaureate students, and postdoctoral fellows. Poster presentations were judged by the keynote speakers as well as TPCB faculty members, with awards sponsored by TPCB and our promotional partners. Notably, the symposium team received important advice on how to stage the online poster session successfully from Dr. Ben Engelhard and Dr. Ali Mohebi, the organizers of the Virtual Dopamine Conference that was held in May 2020.
Early-Stage Students
• Charles Warren (below), UNC Chapel Hill & ChBSP undergraduate (Bachovchin Lab, MSK)
– Using Predictive Tools to Understand the NLRP1 Inflammasome
– ChemBioChem and Nature Chemical Biology Prizes
• Alexa Strauss, TPCB (Levitz Lab, Weill Cornell)
– Domain Coupling in Metabotropic Glutamate Receptors
– Nature Chemical Biology Prize

Two of the award-winning poster presentations, by ChBSP student Charles Warren (left) and TPCB student Adi Berman (right).
TPCB Graduate Students
• Adi Berman (above), TPCB (Kapoor Lab, Rockefeller)
– Characterizing Cellular Functions of the γ-TuRC
– RSC Chemical Biology and TPCB Prizes
• Didar Ciftci, TPCB (Boudker Lab, Weill Cornell)
– Activation Pathways for Faster Aspartate/glutamate Uptake: Lessons Learned from an
Aspartate Transporter
– RSC Chemical Biology and ChemBioChem Prizes
• Chen Chen, TPCB (Heller Lab, MSK)
– Real-time, in vivo Monitoring of Pharmacodynamics of Autophagy Activators Using Organic Color Centers
– Chemical Science Prize
• Ilana Kotliar, TPCB (Sakmar Lab, Rockefeller)
– Multiplexed Analysis of the Secretin-like GPCR-RAMP Interactome by Suspension Bead Array
– Organic & Biomolecular Chemistry Prize


Poster presentations by ChBSP student Joann Chongsaritsinsuk (left) and TPCB student Emily Rundlet (right).
Open Graduate Students
• Jiacheng “Coco” Liu, Weill Cornell (Björn Kafsack Lab)
– Identifying Targets of Histone Reader Domains in Malaria Parasites
– Cell Chemical Biology and TPCB Prizes
• Nicole Williams, CUNY Lehman College (Prabodhika Mallikaratchy Lab)
– Multi Target Ligand-Guided Selection (LIGS) to Generate Aptamers against B-cell
Biomarkers
– Cell Chemical Biology and ChemBioChem Prizes
Senior Scientists
• Lina Freage, CUNY Lehman College (Prabodhika Mallikaratchy Lab)
– Homodimeric Variant of an Aptamer Generated from LIGS Activates TCR-CD3ε Complex
– Nature Chemical Biology and TPCB Prizes


Poster presentation by TPCB student Chloe Burnside (left) and seminar presentation by Prof. Michael Fischbach (right).
The afternoon talks kicked off with a seminar by Prof. Michael Fischbach (above) of Stanford, who described his lab’s recent efforts to build synthetic strain communities of human commensal bacterial, enabling detailed studies of the role of each strain in microbiome metabolism. TPCB student Tandrila Das (below) next spoke about her work with Prof. Howard Hang at Rockefeller to elucidate the role of S-palmitoylation in the antiviral activity and regulation of IFITM3, a protein that is upregulated in the innate immune response, thus disrupting intracellular cholesterol homeostasis. Finally, Prof. Livia Eberlin (below) of the University of Texas at Austin closed out the symposium with an exciting lecture on her development of a MasSpec Pen for in situ, non-destructive assessment of tumor margins during surgical resections, technology that is now being tested in the clinic.

Seminar presentations by TPCB student Tandrila Das (left) and Prof. Livia Eberlin (right).
The symposium continued into the evening with a series of virtual receptions hosted by TPCB students. This year’s event was organized by TPCB students Gabriella Chua, Ilana Kotliar, Lucas Repeta, and Linzhi Ye, who overcame the immense challenges posed by presenting a virtual symposium for the first time. Custom Zoom backgrounds were also created for the event by TPCB student Kristen Vogt. The students were supported by TPCB faculty mentor Prof. Daniel Heller of MSK, TPCB Program Coordinator Margie Hinonangan-Mendoza, and TPCB Director Prof. Derek Tan. The event was broadcast with support from the MSK Department of Marketing & Communications, in particular Jennifer Brosnan, Editorial Production Coordinator, Sam Palmucci, Video Department Manager, and Kim Kennedy. Promotional support was provided by ACS Chemical Biology, Biochemistry, Cell Chemical Biology, ChemBioChem, Chemical Science, Nature Chemical Biology, Organic & Biomolecular Chemistry, RSC Chemical Biology, and the New York Academy of Sciences.
Heeseon An, Viviana Risca, and Ekaterina Vinogradova join the TPCB Faculty
Three new faculty members joined TPCB this summer, bringing new research opportunities in protein engineering, activity-based proteomics, and chromatin structure.
 • Prof. Heeseon An will join the Memorial Sloan Kettering faculty in January 2021. Her lab will use chemical tools to study the quality control mechanisms of subcellular organelles such as ribosomes and lysosomes—two organelles responsible for protein synthesis and degradation. Defects in the maintenance of these organelles are associated with several human genetic diseases. She will use novel protein tags and quantitative proteomics to elucidate the molecular mechanisms underlying the regulation of organelle homeostasis. Prof. An completed her PhD studies in organic chemistry and chemical biology with Prof. Alexander Statsyuk at Northwestern University in 2015, and has carried out her postdoctoral training in cell biology with Prof. J. Wade Harper at Harvard Medical School. [Read more]
• Prof. Heeseon An will join the Memorial Sloan Kettering faculty in January 2021. Her lab will use chemical tools to study the quality control mechanisms of subcellular organelles such as ribosomes and lysosomes—two organelles responsible for protein synthesis and degradation. Defects in the maintenance of these organelles are associated with several human genetic diseases. She will use novel protein tags and quantitative proteomics to elucidate the molecular mechanisms underlying the regulation of organelle homeostasis. Prof. An completed her PhD studies in organic chemistry and chemical biology with Prof. Alexander Statsyuk at Northwestern University in 2015, and has carried out her postdoctoral training in cell biology with Prof. J. Wade Harper at Harvard Medical School. [Read more]
 • Prof. Viviana Risca of The Rockefeller University studies the three-dimensional architecture of chromatin using a technique that she pioneered called RICC-seq (radiation-induced spatially correlated cleavage of DNA with sequencing). She seeks to understand how cells alter chromatin structure during normal processes such as differentation and in response to stressors such as radiation-induced DNA damage or kinase inhibition, thereby controlling transcriptional programs and genome maintenance. Prof. Risca completed her PhD training in biophysics at UC Berkeley in 2015 with Prof. Daniel Fletcher, and her postdoctoral studies in biochemistry and genetics at Stanford with Profs. William J. Greenleaf and Aaron F. Straight. She joined the Rockefeller faculty in 2019. [Read more]
• Prof. Viviana Risca of The Rockefeller University studies the three-dimensional architecture of chromatin using a technique that she pioneered called RICC-seq (radiation-induced spatially correlated cleavage of DNA with sequencing). She seeks to understand how cells alter chromatin structure during normal processes such as differentation and in response to stressors such as radiation-induced DNA damage or kinase inhibition, thereby controlling transcriptional programs and genome maintenance. Prof. Risca completed her PhD training in biophysics at UC Berkeley in 2015 with Prof. Daniel Fletcher, and her postdoctoral studies in biochemistry and genetics at Stanford with Profs. William J. Greenleaf and Aaron F. Straight. She joined the Rockefeller faculty in 2019. [Read more]
 • Prof. Ekaterina (Katya) Vinogradova will join the faculty at The Rockefeller University in October 2020. Her research focuses on elucidating the functions of key proteins that regulate immunological and neuroimmunological processes, using chemical tools and proteomic analyses. In particular, she plans to use electrophilic probes to identify cysteine residues that exhibit different reactivity in physiologic versus pathologic cell states, providing opportunities to target these cysteines selectively in disease systems. Prof. Vinogradova studied organic and organometallic chemistry in her PhD research with Prof. Stephen Buchwald at MIT, graduating in 2015, then continued on to postdoctoral training in chemical biology as a Pfizer Fellow of the Life Sciences Research Foundation with Prof. Benjamin Cravatt at The Scripps Research Institute. [Read more]
• Prof. Ekaterina (Katya) Vinogradova will join the faculty at The Rockefeller University in October 2020. Her research focuses on elucidating the functions of key proteins that regulate immunological and neuroimmunological processes, using chemical tools and proteomic analyses. In particular, she plans to use electrophilic probes to identify cysteine residues that exhibit different reactivity in physiologic versus pathologic cell states, providing opportunities to target these cysteines selectively in disease systems. Prof. Vinogradova studied organic and organometallic chemistry in her PhD research with Prof. Stephen Buchwald at MIT, graduating in 2015, then continued on to postdoctoral training in chemical biology as a Pfizer Fellow of the Life Sciences Research Foundation with Prof. Benjamin Cravatt at The Scripps Research Institute. [Read more]
August 2020
TPCB Welcomes Incoming Class of 2020 with Special Summer Programming
 TPCB welcomed six new students to the program this summer: Clare Cahir (Notre Dame), Sophia Christodolou-Rubalcava (Stanford), Abigail Lemmon (U. Penn.), Victoria Rasmussen (Providence College), Bria VarnBuhler (Pomona College), and Tiffany Zhang (Caltech). While their arrival on campus was delayed by one month due to the COVID-19 pandemic, the students were able to begin their studies on time in early July through specially organized remote programming, completed from their homes across the country. The summer curriculum included weekly Meet the Faculty Seminars presented by TPCB faculty members, journal clubs with students from the Tri-Institutional Chemical Biology Summer Program and with senior TPCB students, and regular research group meetings with their rotation labs. The students also attended a weekly live seminar series sponsored by the MSK Chemical Biology Program, which included student meetings with and presentations by Prof. Heeseon An (MSK incoming faculty), Prof. Paramjit Arora (NYU), Prof. Elizabeth Boon (Stony Brook University), Prof. Ruben Gonzalez (Columbia University), Prof. Mandë Holford (Hunter College), Dr. Luke Lavis (HHMI Janelia Research Campus), Prof. Dirk Trauner (NYU), and Prof. Eranthie Weerapana (Boston College).
TPCB welcomed six new students to the program this summer: Clare Cahir (Notre Dame), Sophia Christodolou-Rubalcava (Stanford), Abigail Lemmon (U. Penn.), Victoria Rasmussen (Providence College), Bria VarnBuhler (Pomona College), and Tiffany Zhang (Caltech). While their arrival on campus was delayed by one month due to the COVID-19 pandemic, the students were able to begin their studies on time in early July through specially organized remote programming, completed from their homes across the country. The summer curriculum included weekly Meet the Faculty Seminars presented by TPCB faculty members, journal clubs with students from the Tri-Institutional Chemical Biology Summer Program and with senior TPCB students, and regular research group meetings with their rotation labs. The students also attended a weekly live seminar series sponsored by the MSK Chemical Biology Program, which included student meetings with and presentations by Prof. Heeseon An (MSK incoming faculty), Prof. Paramjit Arora (NYU), Prof. Elizabeth Boon (Stony Brook University), Prof. Ruben Gonzalez (Columbia University), Prof. Mandë Holford (Hunter College), Dr. Luke Lavis (HHMI Janelia Research Campus), Prof. Dirk Trauner (NYU), and Prof. Eranthie Weerapana (Boston College).
 The new class also completed the Tri-Institutional Responsible Conduct of Research Course and participated in a pilot session of a new discussion focusing on Racism, Discrimination, and Bias in Science along with first-year students from the the Tri-Institutional Computational Biology & Medicine Program. The discussion was led by TPCB Director Prof. Derek Tan, CBM Director Prof. Christina Leslie, Dr. Yaihara Fortis-Santiago (MSK Associate Director of Postdoctoral Affairs and Trainee Diversity Initiatives), and Dr. Avelino Amado (Weill Cornell Diversity Program Manager), with support from Dr. Maika Mitchell (MSK Associate Director of Research Outreach & Compliance), and will serve as the basis for further integration of these important topics into the main Tri-Institutional RCR Course this fall.
The new class also completed the Tri-Institutional Responsible Conduct of Research Course and participated in a pilot session of a new discussion focusing on Racism, Discrimination, and Bias in Science along with first-year students from the the Tri-Institutional Computational Biology & Medicine Program. The discussion was led by TPCB Director Prof. Derek Tan, CBM Director Prof. Christina Leslie, Dr. Yaihara Fortis-Santiago (MSK Associate Director of Postdoctoral Affairs and Trainee Diversity Initiatives), and Dr. Avelino Amado (Weill Cornell Diversity Program Manager), with support from Dr. Maika Mitchell (MSK Associate Director of Research Outreach & Compliance), and will serve as the basis for further integration of these important topics into the main Tri-Institutional RCR Course this fall.
 The three institutions provide extensive resources and support to students, which were introduced in a series of virtual orientation events. Information on the Offices of Career and Professional Development was provided by Dr. Thalyana Stathis (MSK), Dr. Andrea Morris (Rockefeller), and Dr. Aubrey Leukart (Weill Cornell). Important resources for Student Mental Health & Wellness were shared by Dr. Judith Cukor (Weill Cornell), Dr. Nisha Mehta-Naik (Rockefeller), and Dr. Chanchal Sharma (MSK). Further, an overview of Diversity, Inclusion, & Outreach Programs was provided by Dr. Marcus Lambert (Weill Cornell), Dr. Yaihara Fortis-Santiago (MSK), and Dr. Jeanne Garbarino (Rockefeller).
The three institutions provide extensive resources and support to students, which were introduced in a series of virtual orientation events. Information on the Offices of Career and Professional Development was provided by Dr. Thalyana Stathis (MSK), Dr. Andrea Morris (Rockefeller), and Dr. Aubrey Leukart (Weill Cornell). Important resources for Student Mental Health & Wellness were shared by Dr. Judith Cukor (Weill Cornell), Dr. Nisha Mehta-Naik (Rockefeller), and Dr. Chanchal Sharma (MSK). Further, an overview of Diversity, Inclusion, & Outreach Programs was provided by Dr. Marcus Lambert (Weill Cornell), Dr. Yaihara Fortis-Santiago (MSK), and Dr. Jeanne Garbarino (Rockefeller).
The new students were also welcomed to the program through a series of virtual social events organized by the TPCB Orientation Committee of Gabriella Chua, David Hsieh, Gianna Stella, Kristen Vogt, and Lauren Vostal. In addition, TPCB students Chloe Burnside and Chen Chen hosted a new series of biweekly TPCB Ask A Student panels to discuss life in NYC, choosing rotations, impostor syndrome, and choosing a thesis lab. Through the combined efforts of numerous staff, students, and leadership across the three campuses, the first-year students were able to arrive on campus in early August. The students are now embarking on their first laboratory rotations, while benefitting from the academic head start provided by the summer remote curriculum.
ChBSP Students Complete Virtual Summer Internships
 The Tri-Institutional Chemical Biology Summer Program (ChBSP) hosted its third class of outstanding undergraduate students this summer. Due to the COVID-19 pandemic, the internships were all carried out remotely. However, the students were still immersed virtually in TPCB research labs and participated in a wide variety of scientific and career development training activities. The students also joined the TPCB first-year students in a semiweekly journal club led by TPCB students Emma Garst and Natalie Jones, and attended a weekly chemical biology seminar series hosted by the MSK Chemical Biology Program. The ChBSP students presented their work alongside other summer interns in MSK programs at an online poster session on July 31, 2020, showcasing their exposure to forefront research in chemical biology.
The Tri-Institutional Chemical Biology Summer Program (ChBSP) hosted its third class of outstanding undergraduate students this summer. Due to the COVID-19 pandemic, the internships were all carried out remotely. However, the students were still immersed virtually in TPCB research labs and participated in a wide variety of scientific and career development training activities. The students also joined the TPCB first-year students in a semiweekly journal club led by TPCB students Emma Garst and Natalie Jones, and attended a weekly chemical biology seminar series hosted by the MSK Chemical Biology Program. The ChBSP students presented their work alongside other summer interns in MSK programs at an online poster session on July 31, 2020, showcasing their exposure to forefront research in chemical biology.
 Joann Chongsaritsinsuk
Joann Chongsaritsinsuk
University of Richmond, Class of 2021
Structural Basis for Catch Bonding between Mdn1 and Rsa4
with Prof. Tarun Kapoor, Rockefeller
 Samuel Croes
Samuel Croes
Brown University, Class of 2021
Kinome Structure – A Computational Method to Determine Kinase Substrate Specificity
with Prof. Lewis Cantley, Weill Cornell
 Benjamin Freeman
Benjamin Freeman
Princeton University, Class of 2021
Human Embryonic Stem Cells (hESC) for Replacement Therapy and Drug Discovery
with Prof. Shuibing Chen, Weill Cornell
 Anna Soltys
Anna Soltys
Princeton University, Class of 2021
Using Bis-electrophiles to Capture Ub Tetrahedral Intermediates
with Prof. Derek Tan, MSK
 Charles Warren
Charles Warren
University of North Carolina, Chapel Hill, Class of 2021
Exploring NACHT-LRR Binding Proteins’ Influence on Inflammasome Biology
with Prof. Daniel Bachovchin, MSK
ChBSP receives generous financial support from the Sanders Innovation and Education Initiative of the Tri-Institutional Therapeutics Discovery Institute, which is led by Dr. Peter Meinke. The program is directed by TPCB faculty member Prof. Daniel Bachovchin and Dr. Ushma Neill, Vice President of Scientific Education & Training at MSK. [Read more]
June 2020
TPCB Students Continue their Training during the COVID-19 Pandemic
The COVID-19 global pandemic has profoundly impacted everyone’s lives, and TPCB’s graduate students are no exception. The program moved quickly in early March to keep students informed of rapidly evolving policies on the three Tri-Institutional campuses to protect the health and safety of all staff. The three institutions voluntarily paused non-essential laboratory research activities in mid-March, and TPCB transitioned to online enrichment programming to keep students intellectually engaged and socially connected. The members of the TPCB Online Programming Task Force, Nathan Harper, Ilana Kotliar, Elizabeth Orth, Mizuho Horioka, and Linamarie Miller, worked closely with Prof. Derek Tan, TPCB Director and Margie Hinonangan-Mendoza, TPCB Graduate Program Administrator, to organize these activities.
 Over the ensuing months, TPCB students and faculty adapted to these new conditions, working out of their homes, in NYC and abroad, to continue advancing their training and research outside of the laboratory. Classes continued online, with our first-year students completing the TPCB Chemistry in Biology & Medicine course via Zoom meetings. With in-person seminar visits canceled, an online seminar series was established, and TPCB students and faculty came together virtually each week to learn about the latest advances in chemical biology. TPCB students also organized a “Quarantine Journal Club” to discuss recent scientific breakthroughs with their peers. Online lunch meetings and game nights also provided some fun and relaxation for the students. In addition, individual labs organized online group meetings, journal clubs, literature presentations, brainstorming sessions, and social gatherings.
Over the ensuing months, TPCB students and faculty adapted to these new conditions, working out of their homes, in NYC and abroad, to continue advancing their training and research outside of the laboratory. Classes continued online, with our first-year students completing the TPCB Chemistry in Biology & Medicine course via Zoom meetings. With in-person seminar visits canceled, an online seminar series was established, and TPCB students and faculty came together virtually each week to learn about the latest advances in chemical biology. TPCB students also organized a “Quarantine Journal Club” to discuss recent scientific breakthroughs with their peers. Online lunch meetings and game nights also provided some fun and relaxation for the students. In addition, individual labs organized online group meetings, journal clubs, literature presentations, brainstorming sessions, and social gatherings.
Our second-year students worked toward completing their PhD candidacy exams with their thesis committees while our senior students continued to present their work at our weekly Research in Progress Seminar Series. We also welcomed two new TPCB faculty members, Prof. Viviana Risca and Prof. Ekaterina Vinogradova, both of Rockefeller, who presented live seminars on their exciting research programs. The entire TPCB community also came together for four online thesis defenses by Dr. Chaya Stern (Chodera Lab, MSK), Dr. Ashley Chui (Bachovchin Lab, MSK), Dr. Darren Johnson (Bachovchin Lab, MSK), and Dr. Fangyu Liu (Chen Lab, RU), who were surrounded virtually by colleagues, friends, and family.
TPCB also worked to promote mental health awareness throughout the pause, keeping students informed of support resources available within the three institutions and via the NIH Office of Intramural Education and Training. On April 8, Dr. Judith Cukor, Assistant Dean of Student Affairs at Weill Cornell and a clinical psychologist, gave an insightful presentation on “Coping with Changing Routines and Uncertain Times”, sharing helpful strategies for adjusting to working from home. TPCB students also had access to a variety of free, online wellness activities provided by the institutions, including meditation, yoga, and Zumba classes.
As the situation in NYC continued to improve steadily in May and June, laboratories on all three campuses began gradual, staged reopenings, bringing both students and faculty back to the bench to continue their research. Prof. Tan commented, “While the last three months have been incredibly difficult for everyone, I have never been more proud to be part of the TPCB community, and to witness the exceptional resilience and commitment of our students in particular.”
Sahana Rao, Daniel Heller, and Alex Kentsis Recognized at Convocation Ceremonies
TPCB student Sahana Rao and faculty members Prof. Daniel Heller and Prof. Alex Kentsis were recognized with prestigious awards at this year’s academic convocation ceremonies. The events were held virtually due to the COVID-19 pandemic.
 Sahana Rao was honored with the inaugural Marie-Josée Kravis Women in Science Endeavor Graduate Fellowship. The fellowship was recently establshed as part of a broader effort at MSK to recognize the vital importance of recruiting at supporting top female scientists, with the goal of closing the gender gap in research and career opportunities. Sahana’s research focuses on understanding the molecular basis for activation of inflammasomes, innate immune sensors that detect pathogens and endogenous danger signals. Previous work showed that Val-boroPro, a small-molecule inhibitor of dipeptidyl peptidase 8/9 (DPP8/9), induces powerful antitumor responses in syngeneic cancer models by activating the NLRP1 inflammasome in myeloid cells. However, the molecular mechanism of this process remained unclear. Sahana’s thesis research has revealed that, in mice, NLRP1 is activated by autoproteolytic cleavage into two parts, with degradation of one fragment leading to activation of the other. Her current efforts seek to understand the endogenous danger signal that is sensed by the inflammasome as a consequence of DPP8/9 inhibition. Sahana is currently a fourth-year student in the lab of Prof. Daniel Bachovchin at MSK. The Kravis program is made possible by a generous philanthropic gift from Marie-Josée and Henry Kravis. Marie-Josée Kravis is the Chair of the Sloan Kettering Institute and Vice Chair of the MSK Boards of Overseers and Managers. [Read more at MSK: Meet Sahana Rao | Kravis WiSE | Convocation]
Sahana Rao was honored with the inaugural Marie-Josée Kravis Women in Science Endeavor Graduate Fellowship. The fellowship was recently establshed as part of a broader effort at MSK to recognize the vital importance of recruiting at supporting top female scientists, with the goal of closing the gender gap in research and career opportunities. Sahana’s research focuses on understanding the molecular basis for activation of inflammasomes, innate immune sensors that detect pathogens and endogenous danger signals. Previous work showed that Val-boroPro, a small-molecule inhibitor of dipeptidyl peptidase 8/9 (DPP8/9), induces powerful antitumor responses in syngeneic cancer models by activating the NLRP1 inflammasome in myeloid cells. However, the molecular mechanism of this process remained unclear. Sahana’s thesis research has revealed that, in mice, NLRP1 is activated by autoproteolytic cleavage into two parts, with degradation of one fragment leading to activation of the other. Her current efforts seek to understand the endogenous danger signal that is sensed by the inflammasome as a consequence of DPP8/9 inhibition. Sahana is currently a fourth-year student in the lab of Prof. Daniel Bachovchin at MSK. The Kravis program is made possible by a generous philanthropic gift from Marie-Josée and Henry Kravis. Marie-Josée Kravis is the Chair of the Sloan Kettering Institute and Vice Chair of the MSK Boards of Overseers and Managers. [Read more at MSK: Meet Sahana Rao | Kravis WiSE | Convocation]
 Prof. Daniel Heller at MSK was recognized with the 2020 Outstanding Teaching and Mentoring Award from the Weill Cornell Graduate School, which is operated in partership with the Sloan Kettering Institute. The award honors his long-standing commitment to mentoring trainees, including scientists in his own lab and budding biomedical entrepreneurs across the Tri-Institutional campuses. An avid educator, he also co-directs two innovative courses in drug discovery and development, “Drug Development from Molecule to Prescription” and the “Accelerating Bioventure Innovation” entrepreneurship course and training program. Prof. Heller and his laboratory are also active in outreach efforts, including at the Math, Engineering, and Science Academy, a public charter high school in Brooklyn. In fact, Prof. Heller began his career as a 7th and 8th grade science teacher in Houston, Texas. There, he became exposed to chemistry research opportunities at Rice University and the University of Houston, later deciding to pursue his PhD, a postdoc, and ultimately his current faculty position. To date, he has trained over two dozen graduate students and postdoctoral fellows in his lab at MSK. He is known as an open-minded, supportive, and caring mentor who leads by personal example with a positive attitude and infectious enthusiasm. Prof. Heller’s lab develops nanotechnologies to research, diagnose, and treat cancer. His group builds nanoparticles to target drugs to tumors and sensors to detect cancers before they have the chance to spread. They also develop nanoscale tools to interrogate biological processes, such as fluorescent nanosensors that enter the endolysosomal space to detect lipid accumulation within live cells and non-invasively in living organisms. The lab is using these tools to investigate liver diseases and the early-stage genesis of cancers. Prof. Heller is the fourth TPCB faculty member to receive this honor, joining Yueming Li (2018), David Scheinberg (2016), and Derek Tan (2013). [Read more at: Weill Cornell Convocation | Accelerating BioVenture Innovation]
Prof. Daniel Heller at MSK was recognized with the 2020 Outstanding Teaching and Mentoring Award from the Weill Cornell Graduate School, which is operated in partership with the Sloan Kettering Institute. The award honors his long-standing commitment to mentoring trainees, including scientists in his own lab and budding biomedical entrepreneurs across the Tri-Institutional campuses. An avid educator, he also co-directs two innovative courses in drug discovery and development, “Drug Development from Molecule to Prescription” and the “Accelerating Bioventure Innovation” entrepreneurship course and training program. Prof. Heller and his laboratory are also active in outreach efforts, including at the Math, Engineering, and Science Academy, a public charter high school in Brooklyn. In fact, Prof. Heller began his career as a 7th and 8th grade science teacher in Houston, Texas. There, he became exposed to chemistry research opportunities at Rice University and the University of Houston, later deciding to pursue his PhD, a postdoc, and ultimately his current faculty position. To date, he has trained over two dozen graduate students and postdoctoral fellows in his lab at MSK. He is known as an open-minded, supportive, and caring mentor who leads by personal example with a positive attitude and infectious enthusiasm. Prof. Heller’s lab develops nanotechnologies to research, diagnose, and treat cancer. His group builds nanoparticles to target drugs to tumors and sensors to detect cancers before they have the chance to spread. They also develop nanoscale tools to interrogate biological processes, such as fluorescent nanosensors that enter the endolysosomal space to detect lipid accumulation within live cells and non-invasively in living organisms. The lab is using these tools to investigate liver diseases and the early-stage genesis of cancers. Prof. Heller is the fourth TPCB faculty member to receive this honor, joining Yueming Li (2018), David Scheinberg (2016), and Derek Tan (2013). [Read more at: Weill Cornell Convocation | Accelerating BioVenture Innovation]
 Prof. Alex Kentsis received the Louise and Allston Boyer Young Investigator Award for Clinical Research at the MSK Academic Convocation Ceremony on May 27, 2020. This prestigious award honors junior faculty physicians at MSK who have demonstrated great promise and accomplishment in clinical research. Prof. Kentsis is a pediatric oncologist and cancer biologist. His research takes advantage of modern technologies to improve our understanding of the biologic causes of blood and solid tumors. Together with colleagues, he has made discoveries about new therapeutic targets in cancer cells, mechanisms by which cancer cells can evade new treatments, and improved strategies to overcome treatment resistance. Recently, his lab has identified new mechanisms of aberrant gene control and resistance to apoptosis in leukemias, and mechanisms of site-specific oncogenic mutations and DNA repair dependencies in solid tumors. Combined with improved technologies for proteomics and protein engineering, these studies aim to define precise molecular mechanisms that should lead to rational therapeutic strategies for patients. Prof. Kentsis is the first TPCB faculty member to receive the Boyer Clinical Research Award. Five other faculty members have received the Boyer Basic Science Award previously, Morgan Huse (2017), Xuejun Jiang (2007), Christopher Lima (2006), Stephen Long (2016), and Derek Tan (2010). [Read more at: MSK Convocation]
Prof. Alex Kentsis received the Louise and Allston Boyer Young Investigator Award for Clinical Research at the MSK Academic Convocation Ceremony on May 27, 2020. This prestigious award honors junior faculty physicians at MSK who have demonstrated great promise and accomplishment in clinical research. Prof. Kentsis is a pediatric oncologist and cancer biologist. His research takes advantage of modern technologies to improve our understanding of the biologic causes of blood and solid tumors. Together with colleagues, he has made discoveries about new therapeutic targets in cancer cells, mechanisms by which cancer cells can evade new treatments, and improved strategies to overcome treatment resistance. Recently, his lab has identified new mechanisms of aberrant gene control and resistance to apoptosis in leukemias, and mechanisms of site-specific oncogenic mutations and DNA repair dependencies in solid tumors. Combined with improved technologies for proteomics and protein engineering, these studies aim to define precise molecular mechanisms that should lead to rational therapeutic strategies for patients. Prof. Kentsis is the first TPCB faculty member to receive the Boyer Clinical Research Award. Five other faculty members have received the Boyer Basic Science Award previously, Morgan Huse (2017), Xuejun Jiang (2007), Christopher Lima (2006), Stephen Long (2016), and Derek Tan (2010). [Read more at: MSK Convocation]
TPCB Students Present Research at 2020 NYAS Chemical Biology Symposium
The annual New York Academy of Sciences Chemical Biology Symposium on May 21, 2020 was held online this year due to the COVID-19 pandemic. The organizers took advantage of this opportunity to expand the reach of the program, showcasing forefront chemical biology research from around the country. TPCB was well represented, with four students presenting posters, two of whom were selected to present short talks by the organizing committee.
 The day kicked off with a keynote presentation by Prof. Anna Marie Pyle of Yale University, who described her lab’s discovery of novel small-molecule RNA binders that target introns and modulate transcript splicing. This work paves the way to potential therapeutic applications of these molecules in infectious disease and genetic disorders. In one of the student talks, Rachel Leicher (pictured), a TPCB graduate student with Prof. Shixin Liu at Rockefeller, presented her single-molecule studies of the histone methyltransferase complex PRC2, and her discovery that it can bridge non-adjacent nucleosomes, adopting different chromatin binding modes.
The day kicked off with a keynote presentation by Prof. Anna Marie Pyle of Yale University, who described her lab’s discovery of novel small-molecule RNA binders that target introns and modulate transcript splicing. This work paves the way to potential therapeutic applications of these molecules in infectious disease and genetic disorders. In one of the student talks, Rachel Leicher (pictured), a TPCB graduate student with Prof. Shixin Liu at Rockefeller, presented her single-molecule studies of the histone methyltransferase complex PRC2, and her discovery that it can bridge non-adjacent nucleosomes, adopting different chromatin binding modes.
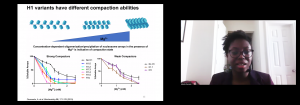 TPCB student Adewola Osunsade (pictured), from the lab of Prof. Yael David at MSK, also shared her recent work on H1 linker histones, in which she developed the first robust approach to the purification of H1s, enabling the biochemical characterization of their functions in chromatin compaction. The second faculty keynote presentation was given by Dr. Elaine Pinheiro of Merck, who described the development of PT2977, now known as MK-6482, as a first-in class Hif-2α inhibitor that has advanced to Phase II human clinical trials in renal cell carcinoma. The compound is a protein–protein interaction inhibitor that acts by blocking heterodimerization of Hif-2α with Hif-1β, thus preventing transcriptional activation of the hypoxia response element.
TPCB student Adewola Osunsade (pictured), from the lab of Prof. Yael David at MSK, also shared her recent work on H1 linker histones, in which she developed the first robust approach to the purification of H1s, enabling the biochemical characterization of their functions in chromatin compaction. The second faculty keynote presentation was given by Dr. Elaine Pinheiro of Merck, who described the development of PT2977, now known as MK-6482, as a first-in class Hif-2α inhibitor that has advanced to Phase II human clinical trials in renal cell carcinoma. The compound is a protein–protein interaction inhibitor that acts by blocking heterodimerization of Hif-2α with Hif-1β, thus preventing transcriptional activation of the hypoxia response element.
The day culminated in an online poster session, which included presentations by representatives from several TPCB labs. Igor Maksimovic, a TPCB student from the lab of Prof. Yael David at MSK, shared his work on the development of new chemical tools to study histone glycation. TPCB student Jonghan Peter Lee, from the lab of Prof. Derek Tan at MSK, also presented his efforts to develop a novel cell-based therapeutic that generates cytotoxic small-molecule drugs locally at the site of a tumor, a project pursued in collaboration with another TPCB faculty member, Prof. David Scheinberg. [Read more]
Congratulations to TPCB’s 2020 Graduates
TPCB congratulates our 2020 graduates, Ashley Chui (Bachovchin Lab, MSK), Alexis Jaramillo Cartagena (Darst Lab, RU), Darren Johnson (Bachovchin Lab, MSK), Jacob Litke (Jaffrey Lab, WCM), Rudolf Pisa (Kapoor Lab, RU), Cristina Santarossa (Kapoor Lab, RU), and Chaya Stern (Chodera Lab, MSK)! They received their PhD degrees at the Weill Cornell Commencement Ceremony on May 28, 2020 and at The Rockefeller University Convocation Ceremony on June 11, 2020. Students in MSK labs were also recognized at the MSK Academic Convocation on May 27, 2020. Notably, three of these students held their thesis defenses online due to the COVID-19 pandemic, surrounded virtually by colleagues, friends, and family. [Read more at: Weill Cornell | Rockefeller (video) | Memorial Sloan Kettering]
 • Ashley Chui, PhD – “N-Terminal Degradation is the Activation Mechanism of the NLRP1B and CARD8 Inflammasomes”
• Ashley Chui, PhD – “N-Terminal Degradation is the Activation Mechanism of the NLRP1B and CARD8 Inflammasomes”
Current position: Postdoctoral Fellow, Prof. Michele Pagano, New York University
 • Alexis Jaramillo Cartagena, PhD – “Structural and Mechanistic Investigations of Crl: An Unconventional Bacterial Activator of RNA Polymerase”
• Alexis Jaramillo Cartagena, PhD – “Structural and Mechanistic Investigations of Crl: An Unconventional Bacterial Activator of RNA Polymerase”
Current position: Postdoctoral Fellow, Dr. Roby Bhattacharyya and Prof. Deborah Hung, Broad Institute
 • Darren Johnson, PhD – “Discovery and Characterization of CARD8-mediated Pyroptosis in Human Blood Cells”
• Darren Johnson, PhD – “Discovery and Characterization of CARD8-mediated Pyroptosis in Human Blood Cells”
Current position: Postdoctoral Fellow, Regeneron Pharmaceuticals, Tarrytown, New York
 • Jacob Litke, PhD – “A Ribozyme-Mediated Mammalian Expression System and its Utility for RNA-Based Devices and Aptamers in Live Cells”
• Jacob Litke, PhD – “A Ribozyme-Mediated Mammalian Expression System and its Utility for RNA-Based Devices and Aptamers in Live Cells”
Current position: Chief Scientific Officer, Chimerna Therapeutics, New York, New York
 • Rudolf Pisa, PhD – “Analyzing Resistance to Design Potent and Selective Inhibitors of AAA Proteins”
• Rudolf Pisa, PhD – “Analyzing Resistance to Design Potent and Selective Inhibitors of AAA Proteins”
Current position: Postdoctoral Fellow, Prof. Tom Rapoport, Harvard Medical School
 • Cristina Santarossa, PhD – “Chemical Biology of Dynein”
• Cristina Santarossa, PhD – “Chemical Biology of Dynein”
Current position: Postdoctoral Fellow, Prof. Gira Bhabha, New York University
 • Chaya Stern, PhD – “Improving Molecular Mechanics Force Fields via Capturing Non-Local, Quantum Effects and Quantifying Parametric Uncertainties”
• Chaya Stern, PhD – “Improving Molecular Mechanics Force Fields via Capturing Non-Local, Quantum Effects and Quantifying Parametric Uncertainties”
Current position: Computational Chemistry Scientist, DeepCure, Boston, Massachusetts
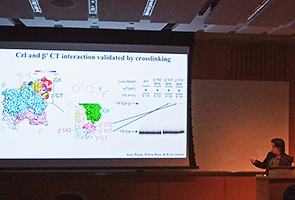

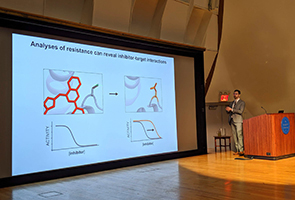


May 2020
Christopher Lima Elected to National Academy of Sciences
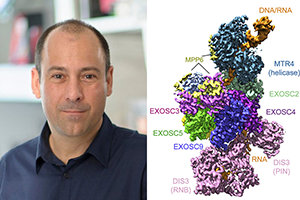 TPCB faculty member Prof. Christopher Lima has been elected to the National Academy of Sciences. Election to the Academy is one of the highest honors in science, and recognizes Prof. Lima’s distinguished contributions in structural biology and biochemistry. His research focuses on elucidating the three-dimensional structures, biochemical mechanisms, and cellular functions of enzymes that are involved in the process of ubiquitin conjugation, a cascading series of reactions that couples ubiquitin to target proteins, thus regulating their activity, stability, subcellular localization, and macromolecular interactions. He is also well-known for his studies of RNA decay pathways that involve the eukaryotic RNA exosome, a large protein complex that directs RNA maturation, processing, and decay. Prof. Lima was one of only 120 scientists across the U.S. elected this year, and joins 9 other TPCB faculty members who have been recognized with this honor. He is also an Investigator of the Howard Hughes Medical Institute and serves as the Chair of the Structural Biology Program at MSK. [Read more at: MSK | National Academy of Sciences]
TPCB faculty member Prof. Christopher Lima has been elected to the National Academy of Sciences. Election to the Academy is one of the highest honors in science, and recognizes Prof. Lima’s distinguished contributions in structural biology and biochemistry. His research focuses on elucidating the three-dimensional structures, biochemical mechanisms, and cellular functions of enzymes that are involved in the process of ubiquitin conjugation, a cascading series of reactions that couples ubiquitin to target proteins, thus regulating their activity, stability, subcellular localization, and macromolecular interactions. He is also well-known for his studies of RNA decay pathways that involve the eukaryotic RNA exosome, a large protein complex that directs RNA maturation, processing, and decay. Prof. Lima was one of only 120 scientists across the U.S. elected this year, and joins 9 other TPCB faculty members who have been recognized with this honor. He is also an Investigator of the Howard Hughes Medical Institute and serves as the Chair of the Structural Biology Program at MSK. [Read more at: MSK | National Academy of Sciences]
March 2020
TPCB Students and Alumni Receive Accolades
Congratulations to TPCB student Ashley Chui and TPCB alumni Dr. Sydney Lu and Dr. Yuan Shi on their recent successes!
 • Ashley Chui has been featured in the MSK Women in Science series in honor of Women’s History Month! Ashley carried out her undergraduate studies at Cal State Fullerton and is currently a graduate student working with Prof. Daniel Bachovchin at MSK. Her thesis research, some of which was published in Science last year, focuses on elucidating the molecular mechanisms by which the NLRP1 and CARD8 inflammasome complexes activate the innate immune system in response to foreign pathogens and small molecules. [Read more at: MSK News]
• Ashley Chui has been featured in the MSK Women in Science series in honor of Women’s History Month! Ashley carried out her undergraduate studies at Cal State Fullerton and is currently a graduate student working with Prof. Daniel Bachovchin at MSK. Her thesis research, some of which was published in Science last year, focuses on elucidating the molecular mechanisms by which the NLRP1 and CARD8 inflammasome complexes activate the innate immune system in response to foreign pathogens and small molecules. [Read more at: MSK News]
 • Dr. Sydney Lu has been awarded a Parker Bridge Fellowship from the Parker Institute for Cancer Immunotherapy! This prestigious fellowship will support Sydney’s ongoing research with Dr. Omar Abdel-Wahab at MSK into the role of disordered RNA splicing in cancer and the development of new potential therapeutics that target this process. Sydney carried out his PhD studies with Prof. Marcel van den Brink at MSK, then completed his MD degree at Stanford Medical School. He returned to MSK in 2015 and is now an Assistant Attending Physician. [Read more at: Parker Institute]
• Dr. Sydney Lu has been awarded a Parker Bridge Fellowship from the Parker Institute for Cancer Immunotherapy! This prestigious fellowship will support Sydney’s ongoing research with Dr. Omar Abdel-Wahab at MSK into the role of disordered RNA splicing in cancer and the development of new potential therapeutics that target this process. Sydney carried out his PhD studies with Prof. Marcel van den Brink at MSK, then completed his MD degree at Stanford Medical School. He returned to MSK in 2015 and is now an Assistant Attending Physician. [Read more at: Parker Institute]
 • Dr. Yuan Shi has been highlighted for his work in drug discovery at Eli Lilly! Yuan graduated from the lab of Prof. David Gin at MSK, researching the total synthesis of the aconitine natural products. He has been working in medicinal chemistry at Lilly for eight years. After starting his career in Indianapolis, where he was promoted to the position of Director, Discovery Chemistry, he recently took on additional responsibilities as Director, Innovation & Partnership in Shanghai, China. [Read more at: Eli Lilly]
• Dr. Yuan Shi has been highlighted for his work in drug discovery at Eli Lilly! Yuan graduated from the lab of Prof. David Gin at MSK, researching the total synthesis of the aconitine natural products. He has been working in medicinal chemistry at Lilly for eight years. After starting his career in Indianapolis, where he was promoted to the position of Director, Discovery Chemistry, he recently took on additional responsibilities as Director, Innovation & Partnership in Shanghai, China. [Read more at: Eli Lilly]
February 2020
TPCB Student Rachel Leicher Receives Biophysical Society Research Award
 TPCB student Rachel Leicher was awarded a Student Research Achievement Award at the Biophysical Society Annual Meeting in San Diego, California on February 17, 2020. The award recognizes Rachel’s work in chromatin biology using single-molecule approaches to study the dynamics and mechanisms of specific epigenetic factors. In particular, she has illustrated how the histone methyltransferase complex PRC2 (polycomb repressive complex 2) engages with long nucleosome substrates to maintain a repressive chromatin state. Using optical tweezers to manipulate nucleosomal arrays, she has obtained mechanical fingerprints of individual PRC2-bound chromatin fibers, uncovering new binding modes that had previously been hidden in biochemical and structural studies. Her work provides a framework for understanding the physical basis of PRC2 recruitment, local chromatin compaction, and epigenetic maintenance. Rachel is currently a graduate student working in the lab of Prof. Shixin Liu at Rockefeller and also received a Biophysical Society Travel Award to attend the conference. [Read more at: Biophysical Society]
TPCB student Rachel Leicher was awarded a Student Research Achievement Award at the Biophysical Society Annual Meeting in San Diego, California on February 17, 2020. The award recognizes Rachel’s work in chromatin biology using single-molecule approaches to study the dynamics and mechanisms of specific epigenetic factors. In particular, she has illustrated how the histone methyltransferase complex PRC2 (polycomb repressive complex 2) engages with long nucleosome substrates to maintain a repressive chromatin state. Using optical tweezers to manipulate nucleosomal arrays, she has obtained mechanical fingerprints of individual PRC2-bound chromatin fibers, uncovering new binding modes that had previously been hidden in biochemical and structural studies. Her work provides a framework for understanding the physical basis of PRC2 recruitment, local chromatin compaction, and epigenetic maintenance. Rachel is currently a graduate student working in the lab of Prof. Shixin Liu at Rockefeller and also received a Biophysical Society Travel Award to attend the conference. [Read more at: Biophysical Society]
December 2019
Vidhya Rangaraju Launches Lab at Max Planck Florida Institute for Neuroscience
 TPCB alumna Vidhya Rangaraju, PhD has launched her independent research program at the Max Planck Florida Institute for Neuroscience in Jupiter, Florida. The overarching goal of her lab is to investigate how neurons, which have unsurpassed morphological complexity, manage mitochondrial energy use and supply. Vidhya completed her graduate training with TPCB faculty member Prof. Timothy Ryan at Weill Cornell, then carried out her postdoctoral studies with Dr. Erin Schuman at the Max Planck Institute for Brain Research in Frankfurt, Germany. She joins nine other TPCB graduates in tenured and tenure-track faculty positions. [Read more at: Max Planck Florida Institute for Neuroscience]
TPCB alumna Vidhya Rangaraju, PhD has launched her independent research program at the Max Planck Florida Institute for Neuroscience in Jupiter, Florida. The overarching goal of her lab is to investigate how neurons, which have unsurpassed morphological complexity, manage mitochondrial energy use and supply. Vidhya completed her graduate training with TPCB faculty member Prof. Timothy Ryan at Weill Cornell, then carried out her postdoctoral studies with Dr. Erin Schuman at the Max Planck Institute for Brain Research in Frankfurt, Germany. She joins nine other TPCB graduates in tenured and tenure-track faculty positions. [Read more at: Max Planck Florida Institute for Neuroscience]
November 2019
David Allis Elected to National Academy of Medicine
 TPCB faculty member Prof. C. David Allis has been elected to the National Academy of Medicine. Founded in 1970 as the Institute of Medicine, election to the Academy is one of the highest honors in medicine and science, recognizing distinguished achievement in a field related to medicine and health. Prof. Allis is a pioneer in the field of epigenetics, and discovered covalent modifications of histone proteins and elucidated their critical roles in the regulation of gene expression and chromatin organization. He is one of 100 new members elected to the Academy this year, joining two other TPCB faculty members, Prof. Lewis Cantley and Prof. Nikola Pavletich. [Read more at: Rockefeller News | National Academy of Medicine]
TPCB faculty member Prof. C. David Allis has been elected to the National Academy of Medicine. Founded in 1970 as the Institute of Medicine, election to the Academy is one of the highest honors in medicine and science, recognizing distinguished achievement in a field related to medicine and health. Prof. Allis is a pioneer in the field of epigenetics, and discovered covalent modifications of histone proteins and elucidated their critical roles in the regulation of gene expression and chromatin organization. He is one of 100 new members elected to the Academy this year, joining two other TPCB faculty members, Prof. Lewis Cantley and Prof. Nikola Pavletich. [Read more at: Rockefeller News | National Academy of Medicine]
October 2019
Three TPCB Faculty Members Awarded Prestigious NIH Grants
The TPCB faculty is supported by over $60 million in research funding annually, providing exceptional resources to support student thesis research in chemical biology. Recently, three TPCB faculty were awarded major NIH grants in recognition of their creativity and impacts in the field. Together, these grants will bring a total of $10 million in additional funding to these labs over the next several years.
 • Prof. Brian Chait at Rockefeller has received a NIH Director’s Transformative Research Award to support his work developing next-generation mass spectrometers that process samples in a massively parallel fashion and will accelerate proteomics research. This award, established in 2009, supports exceptionally innovative, high-risk, and unconventional research projects that have the potential to create or overturn fundamental paradigms or otherwise have unusually broad impact. He is the third TPCB faculty member to receive this award, joining Prof. Samie Jaffrey at Weill Cornell and emeritus Prof. Dinshaw Patel at Sloan Kettering.
• Prof. Brian Chait at Rockefeller has received a NIH Director’s Transformative Research Award to support his work developing next-generation mass spectrometers that process samples in a massively parallel fashion and will accelerate proteomics research. This award, established in 2009, supports exceptionally innovative, high-risk, and unconventional research projects that have the potential to create or overturn fundamental paradigms or otherwise have unusually broad impact. He is the third TPCB faculty member to receive this award, joining Prof. Samie Jaffrey at Weill Cornell and emeritus Prof. Dinshaw Patel at Sloan Kettering.
 • Prof. Samie Jaffrey at Weill Cornell has received a Research Program Award from the National Institute of Neurological Disorders and Stroke to support his studies on the role RNA plays in neurodevelopmental disorders such as Fragile X syndrome, the most common cause of intellectual disability in males. This award provides eight years of funding and is designed to allow established investigators the freedom to embark on ambitious, creative, and longer-term research projects. He is the second TPCB faculty member to receive this award, joining Prof. Vanessa Ruta at Rockefeller. [Read more at: Weill Cornell News]
• Prof. Samie Jaffrey at Weill Cornell has received a Research Program Award from the National Institute of Neurological Disorders and Stroke to support his studies on the role RNA plays in neurodevelopmental disorders such as Fragile X syndrome, the most common cause of intellectual disability in males. This award provides eight years of funding and is designed to allow established investigators the freedom to embark on ambitious, creative, and longer-term research projects. He is the second TPCB faculty member to receive this award, joining Prof. Vanessa Ruta at Rockefeller. [Read more at: Weill Cornell News]
 • Prof. Simon Scheuring at Weill Cornell has received a NIH Director’s Pioneer Award to support the development of new techniques in his lab to record the structural dynamics of ion channels that are activated by physical stimuli, such as force, temperature, or voltage. Since its inception in 2004, this award has supported scientists of exceptional creativity who are pursuing innovative, high-impact approaches to major challenges in biomedical research. He is the second TPCB faculty member to receive this award, joining Prof. Luciano Marraffini at Rockefeller. [Read more at: Weill Cornell News]
• Prof. Simon Scheuring at Weill Cornell has received a NIH Director’s Pioneer Award to support the development of new techniques in his lab to record the structural dynamics of ion channels that are activated by physical stimuli, such as force, temperature, or voltage. Since its inception in 2004, this award has supported scientists of exceptional creativity who are pursuing innovative, high-impact approaches to major challenges in biomedical research. He is the second TPCB faculty member to receive this award, joining Prof. Luciano Marraffini at Rockefeller. [Read more at: Weill Cornell News]
Vanessa Ruta Named 2019 MacArthur Fellow
 TPCB faculty member Prof. Vanessa Ruta has been named a MacArthur Fellow. Colloquially referred to as “genius grants”, the award from the John D. and Catherine T. MacArthur Foundation provides unrestricted support to fellows across a wide range of disciplines in the sciences and arts doing work at the forefront of their fields. Prof. Ruta’s research at Rockefeller University focuses on uncovering the mechanistic basis by which organisms respond to odors. Specifically, she has determined the structure of the first insect odorant receptor and deciphered the neurocircuitry by which flies recognize volatile small-molecule phermomones. Prof. Ruta has spent her entire academic career in New York City, carrying out her undergraduate studies at Hunter College, her PhD at Rockefeller, and her postdoctoral training at Columbia. She joins 26 other individuals who were named this year, which include a criminal justice reformer, theater artist, geochemist, guitarist, urban designer, and poet. [Read more at: Rockefeller News | MacArthur Foundation | CNN | New York Times | NPR | PBS | Washington Post]
TPCB faculty member Prof. Vanessa Ruta has been named a MacArthur Fellow. Colloquially referred to as “genius grants”, the award from the John D. and Catherine T. MacArthur Foundation provides unrestricted support to fellows across a wide range of disciplines in the sciences and arts doing work at the forefront of their fields. Prof. Ruta’s research at Rockefeller University focuses on uncovering the mechanistic basis by which organisms respond to odors. Specifically, she has determined the structure of the first insect odorant receptor and deciphered the neurocircuitry by which flies recognize volatile small-molecule phermomones. Prof. Ruta has spent her entire academic career in New York City, carrying out her undergraduate studies at Hunter College, her PhD at Rockefeller, and her postdoctoral training at Columbia. She joins 26 other individuals who were named this year, which include a criminal justice reformer, theater artist, geochemist, guitarist, urban designer, and poet. [Read more at: Rockefeller News | MacArthur Foundation | CNN | New York Times | NPR | PBS | Washington Post]
September 2019
Forefront Research on Display at the 15th Annual Tri-Institutional Chemical Biology Symposium


Over 150 scientists attended the 15th Annual Tri-Institutional Chemical Biology Symposium on September 4. 2019, including participants from across the NYC area as well as New Jersey, Pennsylvania, and Texas. Prof. Craig Crews of Yale University gave the first faculty keynote address, describing his lab’s pioneering work developing PROTACs (PROteolysis-TArgeting Chimeras), small molecules that induce proteasomal degradation of specific protein targets. TPCB student Linamarie Miller then presented her latest efforts to elucidate the process of ribosome assembly in the lab of Prof. Sebastian Klinge at Rockefeller. TPCB faculty member Prof. Vanessa Ruta, also of Rockefeller, gave the next faculty keynote lecture on her lab’s exciting recent work on the structure and functions of the Orco odorant receptor complex. The morning session closed out with a presentation by TPCB student Darren Johnson on his thesis work with Prof. Daniel Bachovchin at MSK, elucidating the mechanisms and activity of small molecules that induce pyroptotic cell death in acute myeloid leukemia cells.


The lunchtime poster session was again a highlight of the event, with over 50 graduate students, postdoctoral fellows, undergraduates, and high school students presenting their research in chemical biology. Poster prizes were presented in three categories this year, each provided by TPCB’s promotional partners for the symposium, based on judging by the keynote speakers and other faculty members. Early-Stage Student awards went to TPCB students Gabriella Chua (Li Lab, MSK), who received the Chemical Science poster award, and Ilana Kotliar (Sakmar Lab, RU), who received the Chemical Society Reviews poster award. In the Postdoctoral category, Dr. SeCheol Oh (Hite Lab, MSK) was the winner and received a Nature Chemical Biology poster prize. The Graduate Student category saw a two-way tie for second place, with TPCB students Qian Hou (Jaffrey Lab, Weill Cornell) and Adewola Osunsade (David Lab, MSK) both receiving awards from Organic & Biomolecular Chemistry. The outstanding quality of the presentations was exemplified by a four-way tie for first place, with TPCB students Tandrila Das (Hang Lab, RU) and Rudolf Pisa (Kapoor Lab, RU) receiving Cell Chemical Biology poster awards and TPCB student Igor Maksimovic (David Lab, MSK) and Weill Cornell graduate student Vanessa Gutzeit (Levitz Lab, Weill Cornell) receiving prizes from Nature Chemical Biology.


The afternoon session kicked off with a faculty keynote seminar by Prof. Cynthia Wolberger of Johns Hopkins University, who described her lab’s discovery of the intriguing molecular mechanism by which histone H2B ubiquitination and the histone H4 tail organize methylation of a histone H3 lysine by the Dot1L methyltransferase enzyme. The third student lecture was presented by TPCB student Lin Mei, whose work with Prof. Gregory Alushin at Rockefeller involves probing the roles of mechanical force in regulating actin binding by α-catenin, which is important in cellular mechanosensing and mechanotransduction. The final faculty keynote address was given by Prof. Benjamin Cravatt of The Scripps Research Institute, who shared his lab’s latest work using activity-based protein profiling to identify new therapeutic targets.


The symposium concluded with a reception at which the participants continued their discussions of the chemical biology research presented throughout the day. This year’s event was organized by TPCB students Natalie Jones, Jordan Mattheisen, and Nicholas Prescott, working with TPCB faculty member Prof. Daniel Heller.
TPCB Student Tandrila Das Receives FASEB Student Talk Award
 TPCB student Tandrila Das was recognized with the Student Talk Award at the 2019 FASEB Science Research Conference on Protein Lipidation. She presented her recent studies on the mechanism and regulation of the antiviral activity of IFITM (InterFeron-Induced TransMembrane) proteins, which she carried out in the lab of Prof. Howard Hang at Rockefeller. In her talk, entitled “Functional analysis of S-palmitoylated IFITM3 antiviral activity and regulation”, she described her efforts to understand how IFITM3–lipid interactions regulate IFITM3 membrane association and antiviral activity. This year’s conference, entitled “The Protein Lipidation Conference: Enzymology, Signaling, and Therapeutics 2019”, was held at St. Bonaventure University in upstate New York. [Read more at: FASEB]
TPCB student Tandrila Das was recognized with the Student Talk Award at the 2019 FASEB Science Research Conference on Protein Lipidation. She presented her recent studies on the mechanism and regulation of the antiviral activity of IFITM (InterFeron-Induced TransMembrane) proteins, which she carried out in the lab of Prof. Howard Hang at Rockefeller. In her talk, entitled “Functional analysis of S-palmitoylated IFITM3 antiviral activity and regulation”, she described her efforts to understand how IFITM3–lipid interactions regulate IFITM3 membrane association and antiviral activity. This year’s conference, entitled “The Protein Lipidation Conference: Enzymology, Signaling, and Therapeutics 2019”, was held at St. Bonaventure University in upstate New York. [Read more at: FASEB]
August 2019
TPCB Students Attend Annual Career Development Retreat
TPCB students attended the annual TPCB Career Development Retreat this month, spending two days off campus sharing their science and engaging in professional development training and networking. The event, which is organized by TPCB students, was held on August 7–8, 2019 at Crystal Springs Resort in Hamburg, New Jersey. The program began with an ice breaker where the new first-year class was introduced and students played science pictionary. TPCB alumni Dr. Amy Grunbeck Perea (’13, Manager, Immunoassay Platform R&D, Firefly, An Abcam Company) and Dr. John Zinder (’18, Lorraine W. Egan/Damon Runyon Postdoctoral Fellow with Prof. Titia de Lange, Rockefeller University) hosted a panel discussion, sharing their experiences during and after TPCB. Guest speakers Elizabeth DiLuzio and Wheeler del Torro gave a presentation on impostor syndrome.

The Retreat also featured 3-Minute Thesis talks where students were challenged to share their thesis projects in three minutes using a single static slide and a presentation suitable for a broad scientific audience. Ashley Chui (Bachovchin Lab), Linamarie Miller (Klinge Lab), and Rachel Leicher (Liu Lab) were awarded prizes based on voting by their peers. Several senior students also elected to give longer seminars about their research projects to provide a more in-depth discussions. The students also enjoyed social activities including evening karaoke, hiking the Crystal Springs Nature Trail, golfing at the driving range, and swimming and watersliding in the Crystal Springs Biosphere. The event was organized by TPCB students Adi Berman and Elizabeth Orth, with financial support provided by the TPCB NIH T32 Chemistry–Biology Interface Training Grant.



ChBSP Students Cap Off Successful Summer Research Projects
Now in its second year, the Tri-Institutional Chemical Biology Summer Program (ChBSP) hosted another outstanding class of six undergraduate students this summer. The interns conducted chemical biology research in TPCB labs and also took part in scientific, career development, and social activities with other students in Summer at MSK research programs. The students presented their work at the summer poster session on August 8, 2019, which was attended by faculty, postdocs, graduate students, and other scientists across the Tri-Institutional campuses.


ChBSP is directed by TPCB Faculty Member Prof. Daniel Bachovchin and Dr. Ushma Neill, Vice President of Scientific Education & Training at MSK. Over 400 students applied to the program this year, with the 6 interns working on projects across the spectrum of chemical biology at each of the three participating institutions, MSK, Rockefeller University, and Weill Cornell Medicine. The program receives generous financial support from the Sanders Innovation and Education Initiative of the Tri-Institutional Therapeutics Discovery Institute, which is lead by Dr. Peter Meinke. [Read more at: ChBSP]
• Antoinette Antonucci (Kean University) Using Chemical Genetics to Design Selective Chemical Probes of VPS4 (with Prof. Tarun Kapoor, Rockefeller)
• Katharine Kayla Glaser (Barnard College) Expression of ClC-7: A Chloride–Proton Transporter (with Prof. Richard Hite, Sloan Kettering)
• Christopher Nieves Escobar (University of Puerto Rico, Humacao) Structural Analysis of a Lasso Peptide Synthetase (with Prof. Seth Darst, Rockefeller)
• Victoria Rasmussen (Providence College) Discovery of Self-Propargylating Ribozymes in the Pursuit of Self-Methylating RNA (with Prof. Samie Jaffrey, Weill Cornell)
• Matthew See (University of Michigan) Synthesis of Mannose-Phosphate Dolichol (MPD) Analogues to Identify MPD Scramblase (with Prof. Anant Menon, Weill Cornell)
• Shaochen (Tiger) You (University of Texas, Austin) Screening for Inhibitors Using a Coupled Fluorometric Assay (with Prof. Daniel Bachovchin, Sloan Kettering)






July 2019
Structural Studies Shed Light on Molecular Mechanisms of Cystic Fibrosis Drugs
 CFTR (cystic fibrosis transmembrane conductance regulator) is an ion channel that is mutated in cystic fibrosis. These mutations block chloride ion flow through the channel, leading to changes in the mucus lining internal organs. In particular, changes in the lungs cause thick mucus to accumulate, often leading to impaired breathing and persistent infections. Recently, new drugs that potentiate the flow of chloride ions through mutant CFTR have been developed, and one of these, ivacaftor (Kalydeco), was approved by the FDA in 2015. However, because these drugs were discovered through high-throughput screening and empirical medicinal chemistry campaigns, little is known about their mechanisms of action at the molecular level. To address this problem, TPCB student Fangyu Liu and colleagues in the lab of TPCB faculty member Prof. Jue Chen at Rockefeller have determined the cryo-EM structures of ivacaftor and another investigational drug in complex with CFTR. They discovered that both drugs, despite their distinct structures, bind to the same ‘hotspot’ within the transmembrane region of CFTR. This site corresponds with a hinge around which the channel rotates upon ATP binding, suggesting that the drugs may act by stabilizing the open configuration of the pore. This provides detailed structural information that may enable rational design of new CFTR potentiators in the future. The work was published in Science and carried out in collaboration with the lab of Brian Shoichet at UCSF. [Read more at: Rockefeller News | Cystic Fibrosis News Today | Read the article in: Science]
CFTR (cystic fibrosis transmembrane conductance regulator) is an ion channel that is mutated in cystic fibrosis. These mutations block chloride ion flow through the channel, leading to changes in the mucus lining internal organs. In particular, changes in the lungs cause thick mucus to accumulate, often leading to impaired breathing and persistent infections. Recently, new drugs that potentiate the flow of chloride ions through mutant CFTR have been developed, and one of these, ivacaftor (Kalydeco), was approved by the FDA in 2015. However, because these drugs were discovered through high-throughput screening and empirical medicinal chemistry campaigns, little is known about their mechanisms of action at the molecular level. To address this problem, TPCB student Fangyu Liu and colleagues in the lab of TPCB faculty member Prof. Jue Chen at Rockefeller have determined the cryo-EM structures of ivacaftor and another investigational drug in complex with CFTR. They discovered that both drugs, despite their distinct structures, bind to the same ‘hotspot’ within the transmembrane region of CFTR. This site corresponds with a hinge around which the channel rotates upon ATP binding, suggesting that the drugs may act by stabilizing the open configuration of the pore. This provides detailed structural information that may enable rational design of new CFTR potentiators in the future. The work was published in Science and carried out in collaboration with the lab of Brian Shoichet at UCSF. [Read more at: Rockefeller News | Cystic Fibrosis News Today | Read the article in: Science]
Congratulations to TPCB’s 2019 Graduates
TPCB congratulates our 2019 graduates, Shi Chen (Luo Lab, MSK), Zhen Chen (Kapoor Lab, RU), Alejandro Dottore (Kapoor Lab, RU), Zachary Hann (Lima Lab, MSK), and Michaelyn Lux (Tan Lab, MSK). They received their degrees at the Weill Cornell Commencement Ceremony on May 30, 2019 at Carnegie Hall and at The Rockefeller University Convocation Ceremony on June 13, 2019. Students in MSK labs were also recognized at the Gerstner Sloan Kettering Commencement Ceremony on May 29, 2019. [Read more at: Weill Cornell News | Rockefeller News | MSK News]
 • Shi Chen, PhD – “Dynamic Conformational Landscapes and Transition States of Protein Methyltransferases”
• Shi Chen, PhD – “Dynamic Conformational Landscapes and Transition States of Protein Methyltransferases”
Current position: Postdoctoral Fellow, Prof. Squire J. Booker, Pennsylvania State University
 • Zhen Chen, PhD – “Chemical Biology and Structures of Mdn1, an AAA Protein Required for Ribosome Biogenesis”
• Zhen Chen, PhD – “Chemical Biology and Structures of Mdn1, an AAA Protein Required for Ribosome Biogenesis”
Current position: Postdoctoral Fellow, Prof. Ronald Vale, UCSF
 • Alejandro Dottore, PhD – “On the Interactions of Augmin with Microtubules and the Mechanics of the Cross-Linker PRC1”
• Alejandro Dottore, PhD – “On the Interactions of Augmin with Microtubules and the Mechanics of the Cross-Linker PRC1”
Current position: Biopharma Researcher, Buenos Aires, Argentina
 • Zachary Hann, PhD – “Chemical Probes to Trap Ubiquitin Conjugation Cascade Intermediates”
• Zachary Hann, PhD – “Chemical Probes to Trap Ubiquitin Conjugation Cascade Intermediates”
Current position: Postdoctoral Fellow, Prof. Christopher Lima, MSK
 • Michaelyn Lux, PhD – “Development of a Palladium-Catalyzed Bicyclization Reaction and Development of Bis-Electrophilic Probes for Ubiquitin Conjugation”
• Michaelyn Lux, PhD – “Development of a Palladium-Catalyzed Bicyclization Reaction and Development of Bis-Electrophilic Probes for Ubiquitin Conjugation”
Current position: Postdoctoral Fellow, Dr. Charles Yeung, Merck Research Laboratories
June 2019
TPCB Collaboration Reveals Impacts of Conformational Landscapes on Enzyme Function
Enzymes often adopt multiple different conformations in the course of catalyzing chemical reactions. Their conformational preferences can be impacted by mutations, binding of substrates or inhibitors, and interactions with other proteins. This conformational diversity poses challenges in understanding enzyme function and the impacts of disease-associated mutations, and in the design of small-molecule inhibitors. To address this problem, a multidisciplinary team of TPCB researchers has characterized the conformational landscape of an enzyme called SETD8 using a combination of small-molecule inhibitors, biochemical assays, X-ray crystal structures, and massively parallel molecular dynamics simulations, providing striking new insights into these motions. The work was led by TPCB students Shi Chen and Rafal Wiewiora, working in the labs of Prof. Minkui Luo and Prof. John Chodera, respectively, both at MSK, with collaborators at five other institutions.
 SETD8 is a protein lysine methyltransferase enzyme that introduces epigenetic methylation marks on histone tails and is implicated in a variety of cancers. The team first determined a total of four X-ray crystal structures of SETD8, in apo form, bound to one of its substrates S-adenosyl methionine, and bound to two novel inhibitors developed previously in the Luo Lab. Noting distinct conformations in several regions of the enzyme, they used these structures, along with a previously reported structure of SETD8 bound to a histone H4 peptide substrate and a S-adenosyl homocysteine byproduct, to seed thousands of molecular dynamics simulations to identify intermediate conformations and enzyme motions between them. To achieve this, they leveraged the Folding@home network, in which some 33,000 members donated computer processing time for this work, generating an astounding total of six milliseconds of simulations (most simulations span the order of nanoseconds to microseconds). Many cancer-associated mutations of SETD8 are remote from the enzyme active site, making it difficult to rationalize their impacts on enzyme function. Thus, the team next carried out simulations on these SETD8 mutants to understand their impacts on the conformational landscape. This allowed them to predict successfully the activating or inactivating effects of most of these mutations as observed experimentally in biochemical assays. This understanding may now enable chemists to develop drugs to block SETD8 by altering the conformational landscape of this important target. TPCB alumnus Dr. Gil Blum also contributed to the work, which was published last month in eLife. [Read more at: MSK News | Politico | Read the article in: eLife]
SETD8 is a protein lysine methyltransferase enzyme that introduces epigenetic methylation marks on histone tails and is implicated in a variety of cancers. The team first determined a total of four X-ray crystal structures of SETD8, in apo form, bound to one of its substrates S-adenosyl methionine, and bound to two novel inhibitors developed previously in the Luo Lab. Noting distinct conformations in several regions of the enzyme, they used these structures, along with a previously reported structure of SETD8 bound to a histone H4 peptide substrate and a S-adenosyl homocysteine byproduct, to seed thousands of molecular dynamics simulations to identify intermediate conformations and enzyme motions between them. To achieve this, they leveraged the Folding@home network, in which some 33,000 members donated computer processing time for this work, generating an astounding total of six milliseconds of simulations (most simulations span the order of nanoseconds to microseconds). Many cancer-associated mutations of SETD8 are remote from the enzyme active site, making it difficult to rationalize their impacts on enzyme function. Thus, the team next carried out simulations on these SETD8 mutants to understand their impacts on the conformational landscape. This allowed them to predict successfully the activating or inactivating effects of most of these mutations as observed experimentally in biochemical assays. This understanding may now enable chemists to develop drugs to block SETD8 by altering the conformational landscape of this important target. TPCB alumnus Dr. Gil Blum also contributed to the work, which was published last month in eLife. [Read more at: MSK News | Politico | Read the article in: eLife]
Tri-I Chemical Biology Summer Program Welcomes 2019 Interns
 TPCB is pleased to welcome its second class of summer undergraduates to the Chemical Biology Summer Program (ChBSP)! Building upon the success of last summer’s launch, this 10-week internship program gives undergraduates the opportunity to train in TPCB labs doing chemical biology research projects while also participating in seminar series, professional development seminars, and a journal club hosted by current TPCB graduate students. The program will culminate with a poster session on Thursday, August 8, 2019 alongside other summer programs at MSK and Weill Cornell. TPCB faculty member Prof. Daniel Bachovchin directs the program, which is offered through the MSK Office of Scientific Education & Training, led by Dr. Ushma Neill. Generous financial support is being provided by the Sanders Education & Innovation Initiative of the Tri-Institutional Therapeutics Discovery Institute. [Read more at: ChBSP]
TPCB is pleased to welcome its second class of summer undergraduates to the Chemical Biology Summer Program (ChBSP)! Building upon the success of last summer’s launch, this 10-week internship program gives undergraduates the opportunity to train in TPCB labs doing chemical biology research projects while also participating in seminar series, professional development seminars, and a journal club hosted by current TPCB graduate students. The program will culminate with a poster session on Thursday, August 8, 2019 alongside other summer programs at MSK and Weill Cornell. TPCB faculty member Prof. Daniel Bachovchin directs the program, which is offered through the MSK Office of Scientific Education & Training, led by Dr. Ushma Neill. Generous financial support is being provided by the Sanders Education & Innovation Initiative of the Tri-Institutional Therapeutics Discovery Institute. [Read more at: ChBSP]
This year’s ChBSP summer interns are:
- • Antoinette Antonucci, Kean University, Class of 2020, Biotechnology (with Prof. Tarun Kapoor, Rockefeller)
- • Katharine Kayla Glaser, Barnard College, Class of 2020, Biochemistry (with Prof. Richard Hite, Sloan Kettering)
- • Christopher Nieves Escobar, University of Puerto Rico, Humacao, Class of 2021, Chemistry (with Prof. Seth Darst, Rockefeller)
- • Victoria Rasmussen, Providence College, Class of 2020, Biology / Psychology (with Prof. Samie Jaffrey, Weill Cornell)
- • Matthew See, University of Michigan, Class of 2020, Chemistry (with Prof. Anant Menon, Weill Cornell)
- • Shaochen (Tiger) You, University of Texas, Austin, Class of 2020, Biology (with Prof. Daniel Bachovchin, Sloan Kettering)
Annual NYAS Chemical Biology Symposium Features TPCB Students
 The New York Academy of Sciences (NYAS) Chemical Biology Symposium, presented in partnership with TPCB, is an annual highlight in the NYC-area chemical biology community, attended by scientists from across five states. This year’s event on May 22, 2019 featured keynote presentations by Dr. Emma Parmee, VP and Head of Discovery Chemistry at Merck, and Prof. David Spiegel, Professor of Chemistry at Yale. As always, TPCB labs were well-represented by students and postdocs at the event, with many participating in the poster session. TPCB student Ashley Chui was selected for an oral presentation, where she shared her recent work elucidating how N-terminal degradation activates the NLRP1B inflammasome. The work was published recently in Science and carried out with co-first author and TPCB student Sahana Rao and colleagues in the lab of Prof. Daniel Bachovchin at MSK, including TPCB students Darren Johnson and Elizabeth Orth. Postdoctoral fellow Tomasso Cupido, PhD at Rockefeller also presented his recent work on the design of selective chemical inhibitors of AAA proteins, carried out in collaboration with TPCB student Rudolf Pisa and colleagues in the lab of Prof. Tarun Kapoor. TPCB student Didar Ciftci won a F1000 Poster Prize for her poster presentation on single-molecule transport kinetics of a glutamate transporter homologue, carried out in the lab of Prof. Olga Boudker (see image above). [Read more at: NYAS]
The New York Academy of Sciences (NYAS) Chemical Biology Symposium, presented in partnership with TPCB, is an annual highlight in the NYC-area chemical biology community, attended by scientists from across five states. This year’s event on May 22, 2019 featured keynote presentations by Dr. Emma Parmee, VP and Head of Discovery Chemistry at Merck, and Prof. David Spiegel, Professor of Chemistry at Yale. As always, TPCB labs were well-represented by students and postdocs at the event, with many participating in the poster session. TPCB student Ashley Chui was selected for an oral presentation, where she shared her recent work elucidating how N-terminal degradation activates the NLRP1B inflammasome. The work was published recently in Science and carried out with co-first author and TPCB student Sahana Rao and colleagues in the lab of Prof. Daniel Bachovchin at MSK, including TPCB students Darren Johnson and Elizabeth Orth. Postdoctoral fellow Tomasso Cupido, PhD at Rockefeller also presented his recent work on the design of selective chemical inhibitors of AAA proteins, carried out in collaboration with TPCB student Rudolf Pisa and colleagues in the lab of Prof. Tarun Kapoor. TPCB student Didar Ciftci won a F1000 Poster Prize for her poster presentation on single-molecule transport kinetics of a glutamate transporter homologue, carried out in the lab of Prof. Olga Boudker (see image above). [Read more at: NYAS]
TPCB Faculty Members Awarded Pershing Square Sohn Prizes and NIH R35 Grants
TPCB faculty have been recognized with numerous awards to support their forefront research in chemical biology. Most recently, Prof. Yael David and Prof. Alex Kentsis, both at MSK, were awarded the prestigious Pershing Square Sohn Prize for Young Investigators in Cancer Research. The prize provides $600,000 in funding over three years and gives winners the freedom to take risks and pursue their boldest research at a stage when traditional funding is lacking. TPCB faculty members Prof. Daniel Bachovchin and Prof. Daniel Heller have also received this prize previously. [Read more at Pershing Square Sohn: Award Citations and Videos | Press Release]

In addition, four TPCB faculty members have received NIH R35 grants in recognition of their track records of innovative, high-impact research, Prof. Vanessa Ruta and Prof. Tarun Kapoor at Rockefeller, and Prof. Stephen Long and Prof. Minkui Luo at MSK. These grants provide long-term, unrestricted support for their research programs, and are designed to give them the freedom to embark on ambitious, creative projects. Prof. Ruta received a Research Program Award from the National Institute of Neurological Disorders and Stroke, which provides seven years of funding. Profs. Kapoor, Long, and Luo received Maximizing Investigators’ Research Awards (MIRA) from the National Institute of General Medical Sciences, which provide five years of funding. They join six other TPCB faculty members who have received this prestigious type of grant.
May 2019
TPCB Students Ashley Chui, Sahana Rao, and Jacob Litke Win 2019 Rachele Prizes
 Three TPCB students have been awarded the 2019 Julian R. Rachele Prize for their recent research publications. The prize is named for the former Dean of the Weill Cornell Graduate School and his the highest award given to students. The award recognizes publication of a significant research article in the preceding 12-month period, and is typically granted to two students each year. The recipients will present 3-minute overviews of their work at the WCGS Convocation Ceremony on May 29, 2019, and each will also receive a $500 cash prize.
Three TPCB students have been awarded the 2019 Julian R. Rachele Prize for their recent research publications. The prize is named for the former Dean of the Weill Cornell Graduate School and his the highest award given to students. The award recognizes publication of a significant research article in the preceding 12-month period, and is typically granted to two students each year. The recipients will present 3-minute overviews of their work at the WCGS Convocation Ceremony on May 29, 2019, and each will also receive a $500 cash prize.
TPCB students Ashley Chui (left) and Sahana Rao (center) were awarded a joint prize for their work in the lab of Prof. Daniel Bachovchin at Sloan Kettering. Their paper, “N-terminal degradation activates the NLRP1B inflammasome,” was published in the journal Science earlier this year. [Read more at: TPCB News | Read the article in: Science]
TPCB student Jacob Litke (right) was also recognized for his research with Prof. Samie Jaffrey at Weill Cornell. His paper, “Highly efficient expression of circular RNA aptamers in cells using autocatalytic transcripts,” was published in Nature Biotechnology last month. [Read more at: TPCB News | Read the article in: Nat. Biotechnol.]
Jue Chen and Luciano Marraffini Elected to National Academy of Sciences
TPCB is pleased to announce that faculty members Prof. Jue Chen and Prof. Luciano Marraffini, both at Rockefeller, have been elected to the National Academy of Sciences. Members are elected to the Academy in recognition of their distinguished and continuing achievements in original research. Academy membership is a widely accepted mark of excellence in science and is considered one of the highest honors that a scientist can receive. With their elections, a total of nine TPCB faculty members now hold this distinction. [Read more at: Rockefeller News | National Academy of Sciences]
 • Prof. Jue Chen has led the use of cryo-electron microscopy to illuminate the structures and functions of ABC transporters, a family of membrane proteins that pump molecules in or out of cells. These transporters play important roles in many cellular processes, and a better understanding of their activity has broad implications for health and disease. Recent work in her lab with TPCB student Fangyu Liu has shed light on the function and regulation of the CFTR channel, whose mutation results in cystic fibrosis. Prof. Chen joined the TPCB faculty in 2015 after being recruited to Rockefeller from Purdue University. She was also named an HHMI Investigator in 2008.
• Prof. Jue Chen has led the use of cryo-electron microscopy to illuminate the structures and functions of ABC transporters, a family of membrane proteins that pump molecules in or out of cells. These transporters play important roles in many cellular processes, and a better understanding of their activity has broad implications for health and disease. Recent work in her lab with TPCB student Fangyu Liu has shed light on the function and regulation of the CFTR channel, whose mutation results in cystic fibrosis. Prof. Chen joined the TPCB faculty in 2015 after being recruited to Rockefeller from Purdue University. She was also named an HHMI Investigator in 2008.
 • Prof. Luciano Marraffini has carried out pioneering research on CRISPR-Cas9, a bacterial immune system whose adaptation as a gene-editing tool has revolutionized biomedical science and holds promise for the prevention and treatment of many genetic diseases. He showed how this bacterial defense system cuts and destroys invading viral DNA rather than RNA, as was widely believed. Moreover, he was the first to foresee the potential application of CRISPR technology as a programmable gene-editing tool. Prof. Marraffini joined the TPCB faculty in 2012 and was named an HHMI Investigator in 2018.
• Prof. Luciano Marraffini has carried out pioneering research on CRISPR-Cas9, a bacterial immune system whose adaptation as a gene-editing tool has revolutionized biomedical science and holds promise for the prevention and treatment of many genetic diseases. He showed how this bacterial defense system cuts and destroys invading viral DNA rather than RNA, as was widely believed. Moreover, he was the first to foresee the potential application of CRISPR technology as a programmable gene-editing tool. Prof. Marraffini joined the TPCB faculty in 2012 and was named an HHMI Investigator in 2018.
April 2019
TPCB Students Awarded Prestigious NSF Graduate Fellowships
The National Science Foundation (NSF) Graduate Research Fellowship is among the most prestigious and competitive fellowships awarded for graduate study. Since 1952, the program has recognized and supported outstanding graduate students in science, technology, engineering, and mathematics. TPCB is pleased to announce that current student Adi Berman and incoming student Kristen Vogt have received this honor in 2019. In addition, TPCB student Ilana Kotliar received an Honorable Mention in this year’s competition. They join five other current TPCB students who have received NSF fellowships, Natalie Jones, Michaelyn Lux, Adewola Osunsade, Nicholas Prescott, and Chaya Stern. [View the list at: NSF]
 • Adi Berman was awarded a fellowship for her project entitled, “Using Chemical Proteomics to Study Biomolecular Condensation”. The project uses a chemical proteomics approach to profile key protein–protein interactions within biomolecular condensates in live cells. In particular, she will investigate the roles of these interactions in the cell cycle-dependent regulation of the cytoskeleton. Adi is currently a second-year graduate student working with Prof. Tarun Kapoor at Rockefeller.
• Adi Berman was awarded a fellowship for her project entitled, “Using Chemical Proteomics to Study Biomolecular Condensation”. The project uses a chemical proteomics approach to profile key protein–protein interactions within biomolecular condensates in live cells. In particular, she will investigate the roles of these interactions in the cell cycle-dependent regulation of the cytoskeleton. Adi is currently a second-year graduate student working with Prof. Tarun Kapoor at Rockefeller.
 • Kristen Vogt was recognized for her proposal entitled, “Bio-production of synthetic rubber using engineered Escherichia coli”. She is currently a senior at The College of New Jersey and will join TPCB for her graduate studies later this year. She has carried out undergraduate research in computational chemistry with Prof. Joseph Baker and served as President of TCNJ’s chapter of the Gamma Sigma Epsilon chemistry honor society.
• Kristen Vogt was recognized for her proposal entitled, “Bio-production of synthetic rubber using engineered Escherichia coli”. She is currently a senior at The College of New Jersey and will join TPCB for her graduate studies later this year. She has carried out undergraduate research in computational chemistry with Prof. Joseph Baker and served as President of TCNJ’s chapter of the Gamma Sigma Epsilon chemistry honor society.
TPCB Student Jake Litke Creates Perfect Storm for RNA Aptamer Expression
 RNA (ribonucleic acid) plays a central role in translation of the genetically-encoded information in DNA to the cellular functions in proteins. In addition, RNA can fold to form secondary and tertiary structures, and this allows development of short RNA sequences called aptamers that bind specifically to proteins, metabolites, and small molecules. Because they can be genetically encoded, RNA aptamers have exciting applications as live-cell biosensors and inhibitors of cellular pathways. However, this has proven difficult to achieve in mammalian cells due to rapid degradation by exoribonucleases, which trim the ends of the RNA. To overcome this problem, TPCB student Jacob Litke and Prof. Samie Jaffrey at Weill Cornell have developed a highly efficient system for expression of circular RNAs, which are resistant to exoribonucleases. The system, called Tornado (Twister-optimized RNA for durable overexpression), leverages a self-cleaving RNA enzyme called Twister. The RNA aptamer sequence is flanked on both sides by a Twister sequence, leading to cleavage products at either end that are rapidly coupled by an endogenous RNA ligase to form the circular RNA. The system produces circular RNAs 200 times more efficiently than previous systems. The researchers showed that the Tornado system could be used to generate an RNA inhibitor of the NF-κB pathway as well as an intracellular sensor of S-adenosylmethione, a critical enzyme cofactor. The work appeared recently in Nature Biotechnology and Litke was also awarded the 2019 Julian R. Rachele Prize in recognition of his breakthrough research. [Read the article in: Nat. Biotechnol.]
RNA (ribonucleic acid) plays a central role in translation of the genetically-encoded information in DNA to the cellular functions in proteins. In addition, RNA can fold to form secondary and tertiary structures, and this allows development of short RNA sequences called aptamers that bind specifically to proteins, metabolites, and small molecules. Because they can be genetically encoded, RNA aptamers have exciting applications as live-cell biosensors and inhibitors of cellular pathways. However, this has proven difficult to achieve in mammalian cells due to rapid degradation by exoribonucleases, which trim the ends of the RNA. To overcome this problem, TPCB student Jacob Litke and Prof. Samie Jaffrey at Weill Cornell have developed a highly efficient system for expression of circular RNAs, which are resistant to exoribonucleases. The system, called Tornado (Twister-optimized RNA for durable overexpression), leverages a self-cleaving RNA enzyme called Twister. The RNA aptamer sequence is flanked on both sides by a Twister sequence, leading to cleavage products at either end that are rapidly coupled by an endogenous RNA ligase to form the circular RNA. The system produces circular RNAs 200 times more efficiently than previous systems. The researchers showed that the Tornado system could be used to generate an RNA inhibitor of the NF-κB pathway as well as an intracellular sensor of S-adenosylmethione, a critical enzyme cofactor. The work appeared recently in Nature Biotechnology and Litke was also awarded the 2019 Julian R. Rachele Prize in recognition of his breakthrough research. [Read the article in: Nat. Biotechnol.]
March 2019
Ashley Chui and Sahana Rao Elucidate Mechanism that Cells use to Combat Anthrax
 Anthrax is a life-threatening bacterial infection that can be caused by exposure to infected animals and has also been used as an agent of bioterrorism. The bacteria attack the host by releasing virulence factors, one of which is a protease called lethal factor. Lethal factor cleaves and inactivates kinases in the MEK (mitogen-activated protein kinase kinase) family, which is thought to play an important role in compromising the innate immune response to infection. Conversely, lethal factor is also recognized by the innate immune system in mice and rats via its cleavage of inflammasome sensor protein NLRP1B (Nod-like receptor protein 1B; nucleotide-binding domain leucine-rich repeat pyrin domain-containing), which leads to pyroptosis, a form of programmed cell death. However, the molecular mechanism by which this proteolytic cleavage leads to inflammasome activation has remained a mystery. Now, reporting in Science, TPCB students Ashley Chui and Sahana Rao and colleagues in the laboratory of Prof. Daniel Bachovchin at MSK have elucidated this process. The team found that lethal factor cleavage of NLRP1B unveils a destabilizing N-terminal residue that activates proteasomal degradation of the autoinhibitory N-terminal fragment of the protein. This releases the C-terminal fragment, which in turn activates caspase-1, leading to pyroptotic cell death. Intriguingly, they also discovered that small-molecule inhibitors of DPP8/9 (dipeptidyl peptidases 8/9), which the team has investigated as potential anticancer agents, also activate degradation of the NLRP1B N-terminal fragment, but via a distinct pathway. This work sets the stage for further studies of both pathways to understand their roles in pathogen recognition and pyroptotic cell death. TPCB students Darren Johnson and Elizabeth Orth also contributed to the work. [Read more at: MSK News | Read the article in: Science]
Anthrax is a life-threatening bacterial infection that can be caused by exposure to infected animals and has also been used as an agent of bioterrorism. The bacteria attack the host by releasing virulence factors, one of which is a protease called lethal factor. Lethal factor cleaves and inactivates kinases in the MEK (mitogen-activated protein kinase kinase) family, which is thought to play an important role in compromising the innate immune response to infection. Conversely, lethal factor is also recognized by the innate immune system in mice and rats via its cleavage of inflammasome sensor protein NLRP1B (Nod-like receptor protein 1B; nucleotide-binding domain leucine-rich repeat pyrin domain-containing), which leads to pyroptosis, a form of programmed cell death. However, the molecular mechanism by which this proteolytic cleavage leads to inflammasome activation has remained a mystery. Now, reporting in Science, TPCB students Ashley Chui and Sahana Rao and colleagues in the laboratory of Prof. Daniel Bachovchin at MSK have elucidated this process. The team found that lethal factor cleavage of NLRP1B unveils a destabilizing N-terminal residue that activates proteasomal degradation of the autoinhibitory N-terminal fragment of the protein. This releases the C-terminal fragment, which in turn activates caspase-1, leading to pyroptotic cell death. Intriguingly, they also discovered that small-molecule inhibitors of DPP8/9 (dipeptidyl peptidases 8/9), which the team has investigated as potential anticancer agents, also activate degradation of the NLRP1B N-terminal fragment, but via a distinct pathway. This work sets the stage for further studies of both pathways to understand their roles in pathogen recognition and pyroptotic cell death. TPCB students Darren Johnson and Elizabeth Orth also contributed to the work. [Read more at: MSK News | Read the article in: Science]
February 2019
TPCB Student Rudolf Pisa Develops Selective Inhibitors of the AAA Protein Spastin
 Many dynamic cellular processes occur too rapidly to study effectively using classical biological techniques. Enter small-molecule probes, which can provide exquisite temporal control over enzyme functions to dissect their roles in such processes. However, achieving highly selective inhibition of an individual enzyme target can be challenging when there are numerous closely related targets in the cell. Now, in a pair of papers published in Nature Chemical Biology and Journal of the American Chemical Society, TPCB student Rudolf Pisa and colleagues in the laboratory of Prof. Tarun Kapoor at The Rockefeller University have developed novel, selective inhibitors of spastin. This AAA protein (ATPases associated with diverse cellular activities) severs microtubules and plays an important role in cell division and intracellular vesicle transport. There are some 100 AAA proteins that impact a wide range of cellular processes, so developing selective inhibitors is critical to their use as probes. To achieve this, the team homed in on the ATP-binding pocket of spastin and evaluated a series of known kinase inhibitors, which act by binding similarly in the ATP-binding pocket of kinases, to identify compounds that inhibit spastin selectively among a panel of three other AAA proteins. Mutagenesis of active-site residues was then used to identify potential binding interactions, which were further investigated using computational docking and molecular dynamics simulations. The team then leveraged this structural information to design spastazoline, which was a potent spastin inhibitor that did not block any of the other AAA proteins tested nor a large panel of kinases. An X-ray cocrystal structure of a spastazoline analogue bound to spastin further enabled the structure-based design of allele-specific inhibitors using a ‘bump–hole’ strategy. Finally, the team was able to use spastazoline to probe spastin function during cell division, demonstrating its utility as a new chemical probe to dissect this central cellular process. [Read more at: Rockefeller News | Read the articles in: Nat. Chem. Biol. and J. Am. Chem. Soc.]
Many dynamic cellular processes occur too rapidly to study effectively using classical biological techniques. Enter small-molecule probes, which can provide exquisite temporal control over enzyme functions to dissect their roles in such processes. However, achieving highly selective inhibition of an individual enzyme target can be challenging when there are numerous closely related targets in the cell. Now, in a pair of papers published in Nature Chemical Biology and Journal of the American Chemical Society, TPCB student Rudolf Pisa and colleagues in the laboratory of Prof. Tarun Kapoor at The Rockefeller University have developed novel, selective inhibitors of spastin. This AAA protein (ATPases associated with diverse cellular activities) severs microtubules and plays an important role in cell division and intracellular vesicle transport. There are some 100 AAA proteins that impact a wide range of cellular processes, so developing selective inhibitors is critical to their use as probes. To achieve this, the team homed in on the ATP-binding pocket of spastin and evaluated a series of known kinase inhibitors, which act by binding similarly in the ATP-binding pocket of kinases, to identify compounds that inhibit spastin selectively among a panel of three other AAA proteins. Mutagenesis of active-site residues was then used to identify potential binding interactions, which were further investigated using computational docking and molecular dynamics simulations. The team then leveraged this structural information to design spastazoline, which was a potent spastin inhibitor that did not block any of the other AAA proteins tested nor a large panel of kinases. An X-ray cocrystal structure of a spastazoline analogue bound to spastin further enabled the structure-based design of allele-specific inhibitors using a ‘bump–hole’ strategy. Finally, the team was able to use spastazoline to probe spastin function during cell division, demonstrating its utility as a new chemical probe to dissect this central cellular process. [Read more at: Rockefeller News | Read the articles in: Nat. Chem. Biol. and J. Am. Chem. Soc.]
January 2019
TPCB Student Wola Osunsade Develops Robust Method to Prepare H1 Linker Histones
 Compaction of DNA into chromatin plays a critical role in regulating the central cellular process of DNA transcription. Nucleosome core particles, which are comprised of DNA wrapped around an octamer of histone proteins, are further consolidated by the action of H1 linker histones. To date, detailed in vitro studies of H1 histones have been precluded because their C-terminal domains contain long, unstructured, lysine-rich, degradation-prone sequences, making them extremely difficult to isolate and purify. Now, TPCB student Adewola Osunsade and colleagues in the laboratory of Prof. Yael David at MSK have developed a robust method to prepare H1 linker histones. The approach involves expressing H1 histones flanked by two orthogonal tags that promote solubility and proper folding and facilitate purification. The tags are cleaved enzymatically in a traceless fashion to provide native H1 histones. The team was able to use this approach to produce a library of H1 variants, enabling comparisons of their binding affinities to nucleosome core particles. The work provided new insights into the biochemistry of H1 proteins and sets the stage for future analyses of H1 mutations that drive disease through epigenetic modulation of gene transcription. TPCB students Nicholas Prescott, Jakob Hebert, and Devin Ray also contributed to the work, which was published in a special issue of Biochemistry entitled “The Future of Biochemistry: The International Issue”. [Read the article in: Biochemistry]
Compaction of DNA into chromatin plays a critical role in regulating the central cellular process of DNA transcription. Nucleosome core particles, which are comprised of DNA wrapped around an octamer of histone proteins, are further consolidated by the action of H1 linker histones. To date, detailed in vitro studies of H1 histones have been precluded because their C-terminal domains contain long, unstructured, lysine-rich, degradation-prone sequences, making them extremely difficult to isolate and purify. Now, TPCB student Adewola Osunsade and colleagues in the laboratory of Prof. Yael David at MSK have developed a robust method to prepare H1 linker histones. The approach involves expressing H1 histones flanked by two orthogonal tags that promote solubility and proper folding and facilitate purification. The tags are cleaved enzymatically in a traceless fashion to provide native H1 histones. The team was able to use this approach to produce a library of H1 variants, enabling comparisons of their binding affinities to nucleosome core particles. The work provided new insights into the biochemistry of H1 proteins and sets the stage for future analyses of H1 mutations that drive disease through epigenetic modulation of gene transcription. TPCB students Nicholas Prescott, Jakob Hebert, and Devin Ray also contributed to the work, which was published in a special issue of Biochemistry entitled “The Future of Biochemistry: The International Issue”. [Read the article in: Biochemistry]
November 2018
TPCB Student Zhen Chen Uncovers Molecular Mechanism of Ribosome Assembly Protein
 Assembly of cellular ribosomes is a dynamic, multistep process that requires proper organization of ribosomal RNAs and proteins and is coordinated by transient interactions with a host of assembly factors. Mdn1 (midasin) is an AAA protein (ATPase Associated with various Activities) that is essential for proper removal of assembly factors from precursors of the ribosomal 60S subunit. However, Mdn1’s large size (≈5,000 amino acids) and limited homology to other proteins have made it difficult to study its molecular functions. Now, TPCB graduate student Zhen Chen, working in the lab of Prof. Tarun Kapoor at Rockefeller, has determined two cryo-EM structures of Mdn1 that shed light on its mechanism of action. The work builds upon the team’s previous discovery of the Mdn1 inhibitor, Rbin-1. The structures reveal that Mdn1’s MIDAS domain, which binds to ribosome assembly factors, is linked to the catalytic AAA domain via a flexible motif and a striking ≈20 nm structured linker. Moreover, the C-terminal MIDAS domain can dock intramolecularly with the N-terminal AAA ATPase domain in a nucleotide state trapped by the small-molecule inhibitor Rbin-1. Taken together, this study reveals that ATPase-dependent conformational changes in the AAA domain can be transmitted to the MIDAS domain, thus providing a mechanistic connection between ATP hydrolysis and ribosome assembly. This work also highlights the use of a small-molecule inhibitor to dissect the conformational dynamics of an enzyme. The study was published in Cell and carried out in collaboration with the lab of TPCB faculty member Prof. Thomas Walz. [Read more at: Rockefeller News | Read the article in: Cell]
Assembly of cellular ribosomes is a dynamic, multistep process that requires proper organization of ribosomal RNAs and proteins and is coordinated by transient interactions with a host of assembly factors. Mdn1 (midasin) is an AAA protein (ATPase Associated with various Activities) that is essential for proper removal of assembly factors from precursors of the ribosomal 60S subunit. However, Mdn1’s large size (≈5,000 amino acids) and limited homology to other proteins have made it difficult to study its molecular functions. Now, TPCB graduate student Zhen Chen, working in the lab of Prof. Tarun Kapoor at Rockefeller, has determined two cryo-EM structures of Mdn1 that shed light on its mechanism of action. The work builds upon the team’s previous discovery of the Mdn1 inhibitor, Rbin-1. The structures reveal that Mdn1’s MIDAS domain, which binds to ribosome assembly factors, is linked to the catalytic AAA domain via a flexible motif and a striking ≈20 nm structured linker. Moreover, the C-terminal MIDAS domain can dock intramolecularly with the N-terminal AAA ATPase domain in a nucleotide state trapped by the small-molecule inhibitor Rbin-1. Taken together, this study reveals that ATPase-dependent conformational changes in the AAA domain can be transmitted to the MIDAS domain, thus providing a mechanistic connection between ATP hydrolysis and ribosome assembly. This work also highlights the use of a small-molecule inhibitor to dissect the conformational dynamics of an enzyme. The study was published in Cell and carried out in collaboration with the lab of TPCB faculty member Prof. Thomas Walz. [Read more at: Rockefeller News | Read the article in: Cell]
October 2018
Didar Ciftci and Shixin Liu Win Prestigious Research Fellowships
TPCB is pleased to announce two recent research awards to TPCB student Didar Ciftci and TPCB faculty member Prof. Shixin Liu.
 • Didar Ciftci has been awarded an American Heart Association Predoctoral Fellowship. Didar is a 4th-year student pursuing her thesis research with Prof. Olga Boudker at Weill Cornell. This two-year fellowship will support her studies of the mechanism and pharmacologic activation of neurotransmitter glutamate transporters, EAATs. These transporters maintain glutamate concentrations below excito-toxic levels by rapidly pumping the transmitter from the synaptic cleft into neuronal and glial cells in a process powered by electrochemical gradients of ions. Her studies will use a newly developed single-molecule resolution functional assay to study transporter kinetics and the effects of small-molecule probes. This work will expand our fundamental knowledge of the kinetic mechanism of the transporters and lay the foundation for future development of therapeutic EAAT activators.
• Didar Ciftci has been awarded an American Heart Association Predoctoral Fellowship. Didar is a 4th-year student pursuing her thesis research with Prof. Olga Boudker at Weill Cornell. This two-year fellowship will support her studies of the mechanism and pharmacologic activation of neurotransmitter glutamate transporters, EAATs. These transporters maintain glutamate concentrations below excito-toxic levels by rapidly pumping the transmitter from the synaptic cleft into neuronal and glial cells in a process powered by electrochemical gradients of ions. Her studies will use a newly developed single-molecule resolution functional assay to study transporter kinetics and the effects of small-molecule probes. This work will expand our fundamental knowledge of the kinetic mechanism of the transporters and lay the foundation for future development of therapeutic EAAT activators.
 • Prof. Shixin Liu at Rockefeller has received an NIH Director’s New Innovator Award. This 5-year, $2.5 million grant will support his lab’s studies of the asymmetric segregation of histones during DNA replication, which results in different chromatin states in the two daughter strands and plays a pivotal role in cell fate decisions during development and stem cell maintenance. The project aims to develop novel methodologies to probe histone inheritance in a strand-specific manner at the molecular, cellular, and systems levels using single-molecule fluorescence microscopy, super-resolution microscopy, next-generation sequencing, and quantitative mass spectrometry. This multi-pronged approach will establish a framework for characterizing the differential partitioning of histones and for understanding the role of asymmetry in driving epigenome evolution and heterogeneity of cell identity. This work will also pave the way to investigating how the dynamics of epigenetic inheritance are altered in development and disease, providing new therapeutic targets and strategies for developmental disorders, aging, and cancer. Prof. Liu is the eighth TPCB faculty member to receive this prestigious award. [Read more at: NIH]
• Prof. Shixin Liu at Rockefeller has received an NIH Director’s New Innovator Award. This 5-year, $2.5 million grant will support his lab’s studies of the asymmetric segregation of histones during DNA replication, which results in different chromatin states in the two daughter strands and plays a pivotal role in cell fate decisions during development and stem cell maintenance. The project aims to develop novel methodologies to probe histone inheritance in a strand-specific manner at the molecular, cellular, and systems levels using single-molecule fluorescence microscopy, super-resolution microscopy, next-generation sequencing, and quantitative mass spectrometry. This multi-pronged approach will establish a framework for characterizing the differential partitioning of histones and for understanding the role of asymmetry in driving epigenome evolution and heterogeneity of cell identity. This work will also pave the way to investigating how the dynamics of epigenetic inheritance are altered in development and disease, providing new therapeutic targets and strategies for developmental disorders, aging, and cancer. Prof. Liu is the eighth TPCB faculty member to receive this prestigious award. [Read more at: NIH]
September 2018
TPCB Faculty Members David Allis and Samuel Danishefsky Recognized with Major Awards
 TPCB faculty member Prof. C. David Allis of The Rockefeller University has received the 2018 Lasker Award for Basic Medical Research. Widely regarded as the top prize for biomedical research in the United States, the award recognizes Prof. Allis’s seminal discovery of histone acetyltransferases, which introduce acetyl groups onto histone proteins that are associated with chromatin, thus regulating gene transcription. He shares the award with Prof. Michael Grunstein of UCLA, who earlier discovered that histone complexes called nucleosomes regulate transcription and that lysine acetylation on a histone ‘tail’ could activate gene expression. Together, these seminal studies laid the groundwork for the field of epigenetics, which has major implications today in cancer and other human diseases. [Read more at: Rockefeller News | Lasker Foundation]
TPCB faculty member Prof. C. David Allis of The Rockefeller University has received the 2018 Lasker Award for Basic Medical Research. Widely regarded as the top prize for biomedical research in the United States, the award recognizes Prof. Allis’s seminal discovery of histone acetyltransferases, which introduce acetyl groups onto histone proteins that are associated with chromatin, thus regulating gene transcription. He shares the award with Prof. Michael Grunstein of UCLA, who earlier discovered that histone complexes called nucleosomes regulate transcription and that lysine acetylation on a histone ‘tail’ could activate gene expression. Together, these seminal studies laid the groundwork for the field of epigenetics, which has major implications today in cancer and other human diseases. [Read more at: Rockefeller News | Lasker Foundation]
 Prof. Samuel J. Danishefsky, TPCB Emeritus faculty member at the Memorial Sloan Kettering Cancer Center, has been awarded the Sir Derek Barton Gold Medal by the Royal Society of Chemistry. This prestigious award was established in 2001 to commemorate the life and work of Sir Derek Barton, a towering figure in the field of organic chemistry and recipient of the 1969 Nobel Prize in Chemistry. The award recognizes Prof. Danishefsky’s numerous contributions to the art of complex natural product synthesis and the development of new synthetic methods, and their applications to synthetic cancer vaccines and the total chemical synthesis of glycoproteins. Notably, the award specifically recognizes work published by Prof. Danishefsky after the age of 60, which comprises the majority of his tenure at MSK, and includes some 400 papers. This is in keeping with Sir Derek Barton’s major contributions after the mandatory retirement age in the UK, during which time he continued his career at CNRS Paris and later Texas A&M University. Prof. Danishefsky will receive the medal at a small ceremony at MSK in October. [Read more at: Royal Society of Chemistry]
Prof. Samuel J. Danishefsky, TPCB Emeritus faculty member at the Memorial Sloan Kettering Cancer Center, has been awarded the Sir Derek Barton Gold Medal by the Royal Society of Chemistry. This prestigious award was established in 2001 to commemorate the life and work of Sir Derek Barton, a towering figure in the field of organic chemistry and recipient of the 1969 Nobel Prize in Chemistry. The award recognizes Prof. Danishefsky’s numerous contributions to the art of complex natural product synthesis and the development of new synthetic methods, and their applications to synthetic cancer vaccines and the total chemical synthesis of glycoproteins. Notably, the award specifically recognizes work published by Prof. Danishefsky after the age of 60, which comprises the majority of his tenure at MSK, and includes some 400 papers. This is in keeping with Sir Derek Barton’s major contributions after the mandatory retirement age in the UK, during which time he continued his career at CNRS Paris and later Texas A&M University. Prof. Danishefsky will receive the medal at a small ceremony at MSK in October. [Read more at: Royal Society of Chemistry]
Tri-Institutional Chemical Biology Symposium Showcases Research Across NYC
 TPCB presented the 14th Annual Tri-Institutional Chemical Biology Symposium on September 11, 2018 on the campus of The Rockefeller University. The event was attended by some 200 scientists from across the NYC area, including all three Tri-Institutional campuses, NYU, Hunter College, Queens College, the CUNY Advanced Science Research Center, Stony Brook University, and, for the first time, Bard High School Early College. Prof. Squire Booker from Penn State gave the first keynote address describing the role of iron-sulfur cluster proteins in lipoic acid biosynthesis. TPCB student Michaelyn Lux presented her work developing a palladium-catalyzed cascade reaction to synthesize bicyclic ether scaffolds in the Prof. Derek Tan’s lab. Next, Prof. Olga Boudker of Weill Cornell, also a member of the TPCB faculty, shared her latest research on the structural dynamics of glutamate transporter proteins. The morning session closed with a presentation by Jacob Litke, a TPCB student in Prof. Samie Jaffrey’s lab, on a new system for cellular expression of circular RNAs, called Tornado, for use with RNA aptamers and sensors.
TPCB presented the 14th Annual Tri-Institutional Chemical Biology Symposium on September 11, 2018 on the campus of The Rockefeller University. The event was attended by some 200 scientists from across the NYC area, including all three Tri-Institutional campuses, NYU, Hunter College, Queens College, the CUNY Advanced Science Research Center, Stony Brook University, and, for the first time, Bard High School Early College. Prof. Squire Booker from Penn State gave the first keynote address describing the role of iron-sulfur cluster proteins in lipoic acid biosynthesis. TPCB student Michaelyn Lux presented her work developing a palladium-catalyzed cascade reaction to synthesize bicyclic ether scaffolds in the Prof. Derek Tan’s lab. Next, Prof. Olga Boudker of Weill Cornell, also a member of the TPCB faculty, shared her latest research on the structural dynamics of glutamate transporter proteins. The morning session closed with a presentation by Jacob Litke, a TPCB student in Prof. Samie Jaffrey’s lab, on a new system for cellular expression of circular RNAs, called Tornado, for use with RNA aptamers and sensors.



Following lunch, attendees participated in a poster session with over 55 presentations from labs across the NYC area, a threefold increase over last year’s event. Awards were given in seven different categories based on judging by the keynote speakers and TPCB faculty members. In the TPCB student category, Ashley Chui and Sahana Rao, both from the lab of Prof. Daniel Bachovchin, tied for first place with their posters on activation of the Nlrp1b inflammasome, with the third place award going to Zachary Hann in the lab of Prof. Christopher Lima for his presentation on chemical probes of the ubiquitin conjugation cascade. In the TPCB early-stage student and undergraduate category, Chen Chen was awarded first prize for his presentation of his rotation project on leucine transporters with Prof. Boudker. In the final category for postdoctoral fellows and other graduate students, Dr. Eva-Maria Weick placed first for her poster describing her work on the cryo-EM structure of the human RNA exosome–MTR4 complex with Prof. Lima. NYU student Daniel Yoo received the second place award for his presentation on covalent α-helical inhibitors of Ras mutants with Prof. Paramjit Arora. The third place award went to Dr. Sahil Sharma in the lab of TPCB faculty member Prof. Gabriela Chiosis, for his poster on purine-based inhibitors of Grp94, an endoplasmic reticulum Hsp90 paralog. The winners received journal subscription awards generously provided by Cell Chemical Biology, Nature Chemical Biology, and the Royal Society of Chemistry.



Prof. Nieng Yan of Princeton University kicked off the afternoon session with a keynote address on the structures of voltage-gated sodium channels, graciously presenting by Skype from China in the middle of the night as she was unable to attend the symposium in person. TPCB student Fangyu Liu from the lab of Prof. Jue Chen then presented structural insights into the gating mechanism of the CFTR chloride ion channel that is mutated in cystic fibrosis patients. The final keynote presentation of the day was given by Prof. Ronald Raines of MIT, who shared his lab’s latest work leveraging the reactivity of α-diazoketones to label small molecules and proteins. The symposium concluded with a reception for the participants, who continued to discuss the wide range of chemical biology research shared throughout the day.




TPCB received generous support once again this year from its promotional partners, ACS Chemical Biology, Biochemistry, Cell Chemical Biology, Chemical Science, Chemical Society Reviews, Nature Chemical Biology, Organic & Biomolecular Chemistry, and the New York Academy of Sciences. The event was organized by TPCB students Mizuho Horioka, Lin Mei, and Sahana Rao, with guidance from TPCB faculty member Prof. Daniel Heller. [Read more at: Chem. Sci.]
August 2018
Annual TPCB Student Retreat Held at Mohonk Mountain House
 The 2018 TPCB Student Retreat was held on August 7–8, 2018 at Mohonk Mountain House in New Paltz, New York. The retreat was held jointly this year with the Tri-I Computational Biology & Medicine (CBM) PhD program. TPCB and CBM students based at Weill Cornell, Sloan Kettering, Rockefeller University, and Cornell Ithaca were brought together to enjoy two days of networking, scientific development, and relaxation. The Retreat featured three-minute thesis talks by students, career development sessions led by TPCB alumni, and a grantwriting workshop presented by Dr. Ushma Neill, Vice President of Scientific Education & Training at MSK. TPCB alumni Jayakrishnan Nandakumar, PhD (’07, Assistant Professor, University of Michigan) and Nikhil Singla, PhD (’10, Director, Program Management, International AIDS Vaccine Initiative) offered their insights about their respective careers in academia and industrial science. Students also enjoyed social activities including karaoke, trivia, hiking, boating, swimming, and exploring the beautiful Mohonk Mountain House property. The event was organized by TPCB students Jake Hebert and Wola Osunsade, both in the lab of Prof. Yael David at MSK, and was supported by the TPCB NIH T32 Chemistry–Biology Interface Training Grant.
The 2018 TPCB Student Retreat was held on August 7–8, 2018 at Mohonk Mountain House in New Paltz, New York. The retreat was held jointly this year with the Tri-I Computational Biology & Medicine (CBM) PhD program. TPCB and CBM students based at Weill Cornell, Sloan Kettering, Rockefeller University, and Cornell Ithaca were brought together to enjoy two days of networking, scientific development, and relaxation. The Retreat featured three-minute thesis talks by students, career development sessions led by TPCB alumni, and a grantwriting workshop presented by Dr. Ushma Neill, Vice President of Scientific Education & Training at MSK. TPCB alumni Jayakrishnan Nandakumar, PhD (’07, Assistant Professor, University of Michigan) and Nikhil Singla, PhD (’10, Director, Program Management, International AIDS Vaccine Initiative) offered their insights about their respective careers in academia and industrial science. Students also enjoyed social activities including karaoke, trivia, hiking, boating, swimming, and exploring the beautiful Mohonk Mountain House property. The event was organized by TPCB students Jake Hebert and Wola Osunsade, both in the lab of Prof. Yael David at MSK, and was supported by the TPCB NIH T32 Chemistry–Biology Interface Training Grant.
ChBSP Students Present their Summer Research
The Chemical Biology Summer Program (ChBSP) launched this year with an outstanding class of six undergraduate students who spent their summers pursuing forefront chemical biology research in TPCB labs. The students also participated in a wide range of scientific and career development activities with students from other summer undergraduate research programs hosted at MSK. The program culminated with a poster session on August 9, 2018, which was attended by over a hundred scientists from across the Tri-Institutional campuses.


ChBSP Program Director Prof. Daniel Bachovchin commented, “The students accomplished an extraordinary amount of research over the summer, and communicated their findings clearly and thoughtfully at today’s poster session. I could not be more impressed!” ChBSP received generous financial support from the Sanders Innovation & Education Initiative of the Tri-Institutional Therapeutics Discovery Institute (TDI). Dr. Peter Meinke, CEO of TDI, noted, “We recognize programs like ChBSP as key components of the Sanders Innovation and Education Initiative. It is critical that we provide world-class educational opportunities to young scientists, to inspire future generations to aspire to scientific leadership and innovation in new drug discovery and development. The success of this year’s program is a credit to the organizers and an effort that TDI anticipates continuing to support.”
• Angelina Grebe (Fordham University), Progress towards Structural Characterization of Muramyl Dipeptide–NOD2 Complex (with Prof. Howard Hang, Rockefeller)
• Nathan Harper (Lafayette College), Biochemical Analysis of Mdn1, an AAA Protein Required for Ribosome Biogenesis (with Prof. Tarun Kapoor, Rockefeller)
• Sophie Kong (Bucknell University), Initiation of Caspase-1-Dependent Pyroptosis by Activator CARD Domains (with Prof. Daniel Bachovchin, MSK)
• Maham Mahmood (Adelphi University), Screening Modulators that Stabilize the PRMT5–MTA Complex against MTAP/CDKN2A-deleted Cancer Cells (with Prof. Minkui Luo, MSK)
• Rashmi Rao (Cornell University), Structure-Dependent Fluorescence Properties of a Novel Flurogenic Aptamer Derived from Adenine Riboswitch (with Prof. Samie Jaffrey, Weill Cornell)
• Lauren Vostal (Williams College), HIBCH Inhibition in Oncogenic KRAS-drived Pancreatic Ductal Adenocarcinoma (with Prof. John Blenis, Weill Cornell)






TPCB Student Darren Johnson Elucidates Mechanism of Anticancer Drug
 Acute myeloid leukemia (AML) is an aggressive cancer that involves rapid growth of abnormal cells in the bone marrow and blood, interfering with normal development and function of blood cells. Now a team of researchers led by TPCB student Darren Johnson in the lab of Prof. Daniel Bachovchin at MSK has shown that the anticancer drug Val-boroPro (tabalostat) provides a promising new lead for treating AML. Reporting in Nature Medicine, the team found that Val-boroPro, which they have shown previously targets the serine dipeptidases DPP8 and DPP9, has cytotoxicity against both AML cell lines and primary AML cells. They discovered that Val-boroPro activates the protein CARD8, leading to a pro-caspase-1 dependent form of cell death called pyroptosis. Building on these results, they demonstrated in vivo anticancer efficacy of Val-boroPro in mouse models of AML. This work sets the stage for further exploration of DPP8/9 inhibitors as new therapeutics to treat AML. TPCB students Ashley Chui and Sahana Rao, and TPCB faculty member Prof. Alex Kentsis, also contributed to the work. [Read more at: MSK News | Read the article in: Nat. Med.]
Acute myeloid leukemia (AML) is an aggressive cancer that involves rapid growth of abnormal cells in the bone marrow and blood, interfering with normal development and function of blood cells. Now a team of researchers led by TPCB student Darren Johnson in the lab of Prof. Daniel Bachovchin at MSK has shown that the anticancer drug Val-boroPro (tabalostat) provides a promising new lead for treating AML. Reporting in Nature Medicine, the team found that Val-boroPro, which they have shown previously targets the serine dipeptidases DPP8 and DPP9, has cytotoxicity against both AML cell lines and primary AML cells. They discovered that Val-boroPro activates the protein CARD8, leading to a pro-caspase-1 dependent form of cell death called pyroptosis. Building on these results, they demonstrated in vivo anticancer efficacy of Val-boroPro in mouse models of AML. This work sets the stage for further exploration of DPP8/9 inhibitors as new therapeutics to treat AML. TPCB students Ashley Chui and Sahana Rao, and TPCB faculty member Prof. Alex Kentsis, also contributed to the work. [Read more at: MSK News | Read the article in: Nat. Med.]
July 2018
TPCB Congratulates Our 2018 Graduates
Congratulations to the TPCB’s 2018 graduates Malik Chaker-Margot (Klinge Lab, Rockefeller), Adam Trotta (Danishefsky Lab, MSK), and John Zinder (Lima Lab, MSK). Our graduates were recognized at a TPCB luncheon on June 21, 2018. They received their degrees at the Weill Cornell Graduate School graduation ceremony on May 31, 2018 at Carnegie Hall and at The Rockefeller University Convocation Ceremony on June 14, 2018. Students in MSK labs were also recognized at the Gerstner Sloan Kettering Commencement Ceremony on June 1, 2018.
 • Malik Chaker-Margot, PhD – “Making Ribosomes: Biochemical and Structural Studies of Early Ribosomes in Yeast”
• Malik Chaker-Margot, PhD – “Making Ribosomes: Biochemical and Structural Studies of Early Ribosomes in Yeast”
Current position: Postdoctoral Fellow, Prof. Timm Maier, Biozentrum, University of Basel
 • Adam Trotta, PhD – “Towards the Total Synthesis of the Oridamycin and Xiamycin Family of Indolosesquiterpenes”
• Adam Trotta, PhD – “Towards the Total Synthesis of the Oridamycin and Xiamycin Family of Indolosesquiterpenes”
Current position: Postdoctoral Fellow, Prof. Eric Jacobsen, Harvard University
 • John Zinder, PhD – “Structure and Activities of the Saccharomyces cerevisiae Nuclear RNA Exosome”
• John Zinder, PhD – “Structure and Activities of the Saccharomyces cerevisiae Nuclear RNA Exosome”
Current position: Postdoctoral Fellow, Prof. Titia de Lange, The Rockefeller University
Luciano Marraffini Named HHMI Investigator
 TPCB faculty member Prof. Luciano Marraffini of The Rockefeller University has been named a Howard Hughes Medical Institute Investigator. He is one of 19 new HHMI Investigators selected from a pool of 675 applicants for a seven-year term, receiving approximately $8 million in funding. Prof. Marraffini’s lab has performed pioneering studies on how bacteria and archaea defend themselves against viruses. In what has turned out to be a discovery with a remarkably broad array of possible uses, they revealed how a bacterial defense system, CRISPR-Cas, cuts and destroys invading viral DNA rather than RNA strands, as was originally predicted. In collaboration with colleagues at the Broad Institute, Prof. Marraffini reported the first successful genome-editing project based on CRISPR-Cas, technology that has now received significant attention for its potential applications in science and medicine. He joins TPCB’s other five HHMI investigators: Olga Boudker, Jue Chen, Christopher Lima, Roderick MacKinnon, and Nikola Pavletich. [Read more at: Rockefeller News | HHMI]
TPCB faculty member Prof. Luciano Marraffini of The Rockefeller University has been named a Howard Hughes Medical Institute Investigator. He is one of 19 new HHMI Investigators selected from a pool of 675 applicants for a seven-year term, receiving approximately $8 million in funding. Prof. Marraffini’s lab has performed pioneering studies on how bacteria and archaea defend themselves against viruses. In what has turned out to be a discovery with a remarkably broad array of possible uses, they revealed how a bacterial defense system, CRISPR-Cas, cuts and destroys invading viral DNA rather than RNA strands, as was originally predicted. In collaboration with colleagues at the Broad Institute, Prof. Marraffini reported the first successful genome-editing project based on CRISPR-Cas, technology that has now received significant attention for its potential applications in science and medicine. He joins TPCB’s other five HHMI investigators: Olga Boudker, Jue Chen, Christopher Lima, Roderick MacKinnon, and Nikola Pavletich. [Read more at: Rockefeller News | HHMI]
June 2018
TPCB Students and Faculty Recognized with Prestigious Fellowships and Awards
TPCB students and faculty have received numerous fellowships and awards in recognition of their research programs in chemical biology. Recently, TPCB student Chaya Stern and faculty members Prof. Daniel Bachovchin and Prof. Stewart Shuman were honored.
 • Chaya Stern, a 5th year student in the laboratory of Prof. John Chodera at MSK, has been awarded a fellowship from the Molecular Sciences Software Institute to support her work in developing new computational chemistry software. Her project aims to improve the accuracy and quantify the uncertainty of molecular mechanics force fields by generating a database of chemically accurate quantum mechanics data, automating torsion parameter fitting, and propagating force field uncertainties to predictions. The fully open-source software is being designed for the molecular simulation community and is part of the Open Forcefield Initiative. [Read more at: MolSSI]
• Chaya Stern, a 5th year student in the laboratory of Prof. John Chodera at MSK, has been awarded a fellowship from the Molecular Sciences Software Institute to support her work in developing new computational chemistry software. Her project aims to improve the accuracy and quantify the uncertainty of molecular mechanics force fields by generating a database of chemically accurate quantum mechanics data, automating torsion parameter fitting, and propagating force field uncertainties to predictions. The fully open-source software is being designed for the molecular simulation community and is part of the Open Forcefield Initiative. [Read more at: MolSSI]
 • Prof. Daniel Bachovchin at MSK has been recognized with two prestigious fellowships for junior faculty. He was one of seven scientists to receive the Pershing Square Sohn Prize for Young Investigators in Cancer Research. His project aims to develop a highly innovative chemical-proteomics platform to determine the mechanism of action of anti-cancer protease inhibitors. He is the second TPCB faculty member to receive this honor, following Prof. Daniel Heller who was recognized last year. In addition, Prof. Bachovchin has been named a 2018 Sloan Research Fellow in Chemistry. This award recognizes his work in chemical biology as one of the most promising early-career scientists in the U.S. and Canada. Four other TPCB faculty have received this distinction previously: Prof. Sebastian Klinge (2014), Prof. Vanessa Ruta (2013), Prof. Derek Tan (2007), and Prof. Timothy Ryan (1999). [Read more at: Pershing Square Sohn | Alfred P. Sloan Foundation]
• Prof. Daniel Bachovchin at MSK has been recognized with two prestigious fellowships for junior faculty. He was one of seven scientists to receive the Pershing Square Sohn Prize for Young Investigators in Cancer Research. His project aims to develop a highly innovative chemical-proteomics platform to determine the mechanism of action of anti-cancer protease inhibitors. He is the second TPCB faculty member to receive this honor, following Prof. Daniel Heller who was recognized last year. In addition, Prof. Bachovchin has been named a 2018 Sloan Research Fellow in Chemistry. This award recognizes his work in chemical biology as one of the most promising early-career scientists in the U.S. and Canada. Four other TPCB faculty have received this distinction previously: Prof. Sebastian Klinge (2014), Prof. Vanessa Ruta (2013), Prof. Derek Tan (2007), and Prof. Timothy Ryan (1999). [Read more at: Pershing Square Sohn | Alfred P. Sloan Foundation]
 • Prof. Stewart Shuman at MSK has received an NIH Maximizing Investigators’ Research (MIRA) Award. These extended, 5-year grants were established by the National Institute of General Medical Sciences (NIGMS) in 2015 to provide investigators with greater funding stability and scientfiic flexibility, enhancing the chances for important breakthroughs. Prof. Shuman’s long-standing and highly-accomplished research program seeks to illuminate fundamental DNA and RNA transactions at a molecular level. His lab studies the mechanisms, structures, and regulation of enzymes that perform nucleic acid synthesis, modification, and repair. Current targets include polynucleotide ligases, mRNA capping enzymes, tRNA splicing enzymes, ribotoxin, and the RNA polymerase II C-terminal domain. He is the fifth TPCB faculty member to receive NIGMS MIRA support, joining Prof. Sean Brady, Prof. Seth Darst, Prof. Joshua Levitz (Early-Stage Investigator Award), and , Prof. Christopher Lima.
• Prof. Stewart Shuman at MSK has received an NIH Maximizing Investigators’ Research (MIRA) Award. These extended, 5-year grants were established by the National Institute of General Medical Sciences (NIGMS) in 2015 to provide investigators with greater funding stability and scientfiic flexibility, enhancing the chances for important breakthroughs. Prof. Shuman’s long-standing and highly-accomplished research program seeks to illuminate fundamental DNA and RNA transactions at a molecular level. His lab studies the mechanisms, structures, and regulation of enzymes that perform nucleic acid synthesis, modification, and repair. Current targets include polynucleotide ligases, mRNA capping enzymes, tRNA splicing enzymes, ribotoxin, and the RNA polymerase II C-terminal domain. He is the fifth TPCB faculty member to receive NIGMS MIRA support, joining Prof. Sean Brady, Prof. Seth Darst, Prof. Joshua Levitz (Early-Stage Investigator Award), and , Prof. Christopher Lima.
TPCB’s Chemical Biology Summer Program Welcomes Inaugural Class
 TPCB is pleased to welcome the inaugural class of undergraduate summer interns to the new Chemical Biology Summer Program (ChBSP)! This 10-week program gives undergraduate students the opportunity to pursue forefront research in chemical biology in TPCB labs, working alongside our graduate students and faculty. ChBSP students also attend scientific lectures and professional development seminars as part of their summer experience. The program will culminate with a poster session on Thursday, August 9th, showcasing summer undergraduate research from across the Tri-Institutional campuses. [Read more at: ChBSP]
TPCB is pleased to welcome the inaugural class of undergraduate summer interns to the new Chemical Biology Summer Program (ChBSP)! This 10-week program gives undergraduate students the opportunity to pursue forefront research in chemical biology in TPCB labs, working alongside our graduate students and faculty. ChBSP students also attend scientific lectures and professional development seminars as part of their summer experience. The program will culminate with a poster session on Thursday, August 9th, showcasing summer undergraduate research from across the Tri-Institutional campuses. [Read more at: ChBSP]
This year’s ChBSP summer interns are:
- • Angelina Grebe, Fordham University, Class of 2019, Chemistry (with Prof. Howard Hang, Rockefeller)
- • Nathan Harper, Lafayette College, Class of 2019, Biology (with Prof. Tarun Kapoor, Rockefeller)
- • Sophie Kong, Bucknell University, Class of 2019, Biochemistry (with Prof. Daniel Bachovchin, MSK)
- • Maham Mahmood, Adelphi University, Class of 2019, Biochemistry (with Minkui Luo, MSK)
- • Rashmi Rao, Cornell University, Class of 2020, Chemistry (with Prof. Samie Jaffrey, Weill Cornell)
- • Lauren Vostal, Williams College, Class of 2019, Chemistry (with Prof. John Blenis, Weill Cornell)
May 2018
TPCB Research Features at NYAS 2018 Chemical Biology Symposium
The New York Academy of Sciences (NYAS) annual year-end Chemical Biology Symposium, presented in partnership with TPCB, is a highlight for chemical biologists across the tristate area, bringing together undergraduates, graduate students, postdocs, faculty, and staff scientists from numerous major academic institutions and pharmaceutical companies. This year’s event was held on May 23, 2018 and featured a keynote presentation by Prof. Joanne Stubbe of MIT. Prof. Stubbe described her long-standing research program on the structure and function of ribonucleotide reductase, with recent efforts focusing on elucidating striking conformational changes that regulate long-range, proton-coupled electron transfer from an iron center to the ribonucleotide substrate. Students and postdocs from TPCB labs were well-represented in the poster session, and several were selected for oral presentations, including TPCB student Jacob Litke, who presented his work with Prof. Samie Jaffrey at Weill Cornell on developing a platform for efficient expression of circular RNA sensors; postdoctoral fellow Dr. Cornelius Taabazuing of Prof. Dan Bachovchin’s lab at MSK, who described his discovery of crosstalk between pyroptotic and apoptotic signaling pathways, and recent graduate, Dr. Zhenrun (Jerry) Zhang of Prof. Howard Hang’s lab at Rockefeller, who discussed the role of short-chain fatty acids in Salmonella virulence. In addition, TPCB student Michaelyn Lux, from Prof. Derek Tan’s lab at MSK, was awarded one of three F1000 poster prizes for her presentation on the development of a new palladium cascade reaction to generate fused bicyclic ethers. [Read more at: NYAS]
April 2018
TPCB Student Sahana Rao Uncovers Mechanism of Novel Anticancer Agent
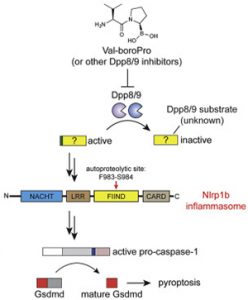 Val-boroPro is a small molecule that triggers a form of programmed cell death called pyroptosis in certain cell types and induces immune-mediated tumor regression in mouse models of cancer. Now, reporting in Cell Chemical Biology, TPCB student Sahana Rao and coworkers in the laboratory of Prof. Daniel Bachovchin at Sloan Kettering present new insights into the mechanism of pyroptosis induction. Previous work in the Bachovchin lab demonstrated that Val-boroPro acts by inhibiting the dipeptidyl peptidases DPP8 and DPP9, but the mechanism by which this induces pyroptotic cell death was unknown. Using Val-boroPro as a chemical probe, they now show that this inhibition activates the Nlrp1b inflammasome, a protein complex that promotes cellular inflammatory responses leading to cytokine secretion and cell death via pyroptosis. TPCB students Ashley Chui and Darren Johnson in the Bachovchin lab also contributed. This study reveals a new mechanism for activation of the innate immune response and suggests a role for DPP8/9 in regulation of the innate immune system. [Read the article in: Cell Chem. Biol.]
Val-boroPro is a small molecule that triggers a form of programmed cell death called pyroptosis in certain cell types and induces immune-mediated tumor regression in mouse models of cancer. Now, reporting in Cell Chemical Biology, TPCB student Sahana Rao and coworkers in the laboratory of Prof. Daniel Bachovchin at Sloan Kettering present new insights into the mechanism of pyroptosis induction. Previous work in the Bachovchin lab demonstrated that Val-boroPro acts by inhibiting the dipeptidyl peptidases DPP8 and DPP9, but the mechanism by which this induces pyroptotic cell death was unknown. Using Val-boroPro as a chemical probe, they now show that this inhibition activates the Nlrp1b inflammasome, a protein complex that promotes cellular inflammatory responses leading to cytokine secretion and cell death via pyroptosis. TPCB students Ashley Chui and Darren Johnson in the Bachovchin lab also contributed. This study reveals a new mechanism for activation of the innate immune response and suggests a role for DPP8/9 in regulation of the innate immune system. [Read the article in: Cell Chem. Biol.]
March 2018
TPCB Student Linamarie Miller Elucidates Maturation of Ribosome Large Subunit
 The ribosome is built from proteins and ribosomal RNAs that are assembled and folded in a complex, multistage process. In a new report in Nature, TPCB student Linamarie Miller and colleagues in the laboratory of Prof. Sebastian Klinge at Rockefeller describe the cryo-EM structures of the nucleolar pre-60S ribosomal subunit in three different conformational states. Some 21 ribosome assembly factors work together to stabilize and remodel the rRNA and protein constituents. The work was performed in collaboration with the lab of TPCB faculty member Prof. Brian Chait, also at Rockefeller, who carried out cross-linking and mass spectrometry studies to help characterize the architecture of the assembly factors. TPCB student Malik Chaker-Margot in the Klinge lab also contributed. This work provides new insights into the molecular mechanisms that drive ribosome assembly forward in a unidirectional manner. [Read more at: Rockefeller News | Read the article in: Nature]
The ribosome is built from proteins and ribosomal RNAs that are assembled and folded in a complex, multistage process. In a new report in Nature, TPCB student Linamarie Miller and colleagues in the laboratory of Prof. Sebastian Klinge at Rockefeller describe the cryo-EM structures of the nucleolar pre-60S ribosomal subunit in three different conformational states. Some 21 ribosome assembly factors work together to stabilize and remodel the rRNA and protein constituents. The work was performed in collaboration with the lab of TPCB faculty member Prof. Brian Chait, also at Rockefeller, who carried out cross-linking and mass spectrometry studies to help characterize the architecture of the assembly factors. TPCB student Malik Chaker-Margot in the Klinge lab also contributed. This work provides new insights into the molecular mechanisms that drive ribosome assembly forward in a unidirectional manner. [Read more at: Rockefeller News | Read the article in: Nature]
February 2018
TPCB Alumni Launch Independent Research Programs
Graduate training in TPCB prepares students for a wide range of scientific careers. Among our alumni are 9 tenure-track faculty, including Doeke Hekstra, Rashad Karimov, Sumana Sanyal, and Qian Yin who recently launched their independent labs:
 • Doeke Hekstra, PhD (Assistant Professor, Department of Molecular & Cellular Biology, Harvard University) studies the mechanics of biomolecules, and more generally the role of physical and evolutionary forces in shaping biological systems. [Read more at: Harvard]
• Doeke Hekstra, PhD (Assistant Professor, Department of Molecular & Cellular Biology, Harvard University) studies the mechanics of biomolecules, and more generally the role of physical and evolutionary forces in shaping biological systems. [Read more at: Harvard]
 • Rashad Karimov, PhD (Assistant Professor, Department of Chemistry & Biochemistry, Auburn University) develops new methodologies for the synthesis of saturated nitrogen heterocycles, involving dearomatization of nitrogen heteroaromatics and new hetero-cycloaddition and annulation reactions. [Read more at: Auburn]
• Rashad Karimov, PhD (Assistant Professor, Department of Chemistry & Biochemistry, Auburn University) develops new methodologies for the synthesis of saturated nitrogen heterocycles, involving dearomatization of nitrogen heteroaromatics and new hetero-cycloaddition and annulation reactions. [Read more at: Auburn]
 • Sumana Sanyal, PhD (Assistant Professor, Division of Public Health Laboratory Sciences, Pasteur Research Pole, The University of Hong Kong) focuses on understanding the role of ubiquitin and ubiquitin-like modifiers in host–pathogen interactions that orchestrate an outcome between successful viral infection versus host restriction. [Read more at: HKU Pasteur]
• Sumana Sanyal, PhD (Assistant Professor, Division of Public Health Laboratory Sciences, Pasteur Research Pole, The University of Hong Kong) focuses on understanding the role of ubiquitin and ubiquitin-like modifiers in host–pathogen interactions that orchestrate an outcome between successful viral infection versus host restriction. [Read more at: HKU Pasteur]
 • Qian Yin, PhD (Assistant Professor, Department of Biological Science, Florida State University) studies the function of IFN-inducible GTPases and their activation and regulatory mechanisms in inflammation, autophagy, and host–pathogen interactions using biochemical, structural, cellular, and pharmacological approaches. [Read more at: Florida State]
• Qian Yin, PhD (Assistant Professor, Department of Biological Science, Florida State University) studies the function of IFN-inducible GTPases and their activation and regulatory mechanisms in inflammation, autophagy, and host–pathogen interactions using biochemical, structural, cellular, and pharmacological approaches. [Read more at: Florida State]
November 2017
TPCB Launches Summer Undergraduate Internship Program
TPCB is excited to announce the launch of a new Chemical Biology Summer Program (ChBSP) for undergraduates in the New York City area. This 10-week program will launch in the summer of 2018, giving students the opportunity to perform cutting-edge research in chemical biology in the laboratory of a TPCB faculty member. Students will work alongside graduate students and postdocs in the labs and will also attend weekly research seminars presented by the faculty and workshops on professional and career development. Students will present their research in a poster session at the end of the summer, alongside students from a broad range of other summer programs hosted on the Tri-Institutional campuses.
The program is directed by TPCB faculty member Prof. Daniel Bachovchin at Sloan Kettering and is receiving generous financial support from the Sanders Educational Initiative of the Tri-Institutional Therapeutics Discovery Institute and organizational support from the MSK Office of Education and Training. Students will receive a $4,000 stipend. However, no housing is provided, so the program is geared toward students who already have local accommodations in the NYC area. US citizenship or permanent residency is required. Online applications are open from November 1, 2017 to February 2, 2018. [Read more at: ChBSP]
September 2017
TPCB Presents the 13th Annual Tri-Institutional Chemical Biology Symposium
 The 13th Annual Chemical Biology Symposium, presented by TPCB, was held on September 7, 2017 at MSK. Organized by TPCB students Ashley Chui, Rachel Leicher, and Linamarie Miller, the event was attended by over 150 scientists from across the greater NYC area. The day started with a faculty keynote lecture by Prof. Helen Blackwell from the University of Wisconsin–Madison, who presented her lab’s work on small-molecule probes of bacterial quorum sensing systems. TPCB student John Zinder shared his research on characterizing the structure and function of the RNA exosome in the lab of Prof. Christopher Lima at MSK. TPCB faculty member Prof. Daniel Heller gave the second keynote lecture on nanochemical biology approaches to drug delivery and molecular imaging. The morning session concluded with a talk by TPCB student Shi Chen, who described his work on the conformational dynamics of the protein lysine methyltransferase SET8, carried out in the lab of Prof. Minkui Luo in collaboration with TPCB student Rafal Wiewora in the lab of Prof. John Chodera at MSK.
The 13th Annual Chemical Biology Symposium, presented by TPCB, was held on September 7, 2017 at MSK. Organized by TPCB students Ashley Chui, Rachel Leicher, and Linamarie Miller, the event was attended by over 150 scientists from across the greater NYC area. The day started with a faculty keynote lecture by Prof. Helen Blackwell from the University of Wisconsin–Madison, who presented her lab’s work on small-molecule probes of bacterial quorum sensing systems. TPCB student John Zinder shared his research on characterizing the structure and function of the RNA exosome in the lab of Prof. Christopher Lima at MSK. TPCB faculty member Prof. Daniel Heller gave the second keynote lecture on nanochemical biology approaches to drug delivery and molecular imaging. The morning session concluded with a talk by TPCB student Shi Chen, who described his work on the conformational dynamics of the protein lysine methyltransferase SET8, carried out in the lab of Prof. Minkui Luo in collaboration with TPCB student Rafal Wiewora in the lab of Prof. John Chodera at MSK.
Symposium participants were then treated to lunch and a poster session highlighting recent advances in chemical biology research across the Tri-Institutional Research Program. Poster prizes were awarded to TPCB students Rudolph Pisa (Kapoor Lab, RU), Darren Johnson (Bachovchin Lab, MSK), and Michaelyn Lux (Tan Lab, MSK) and postdoctoral fellow Nathan Westcott (Hang Lab, RU). Journal subscription awards were generously sponsored by the Royal Society of Chemistry, Cell Chemical Biology, and Nature Chemical Biology, and the winners also received gift cards from TPCB.




In the afternoon session, Prof. Dorothee Kern of Brandeis University gave an engaging and music-filled presentation on her lab’s studies of the evolution of enzyme catalysis and regulation. TPCB student Zhen Chen then shared his research in the lab of Prof. Tarun Kapoor at RU on the discovery of novel ribosome biogenesis inhibitors that target the AAA protein Mdn1. Prof. Kevan Shokat of UCSF gave the final keynote presentation on his lab’s recent progress developing novel small-molecule inhibitors of mTOR and KRas for cancer therapy. The event concluded with a reception at which participants continued to discuss the latest advances in the field over food and drinks.
TPCB received generous support for the event from our promotional partners, ACS Chemical Biology, Biochemistry, Cell Chemical Biology, Chemical Science, Chemical Society Reviews, Nature Chemical Biology, Organic & Biomolecular Chemistry, and the New York Academy of Sciences. [Read more at: Org. Biomol. Chem.]
August 2017
Students Share Research and Get Career Advice from Alumni at 2017 TPCB Retreat
The 2017 TPCB Student Retreat was held on August 14-15, 2017 at Mohonk Mountain House, New Paltz, NY. The event brought all TPCB students based at Weill Cornell, Sloan Kettering and Rockefeller University together for the summer. The retreat featured research talks by students in the form of three-minute thesis presentations and career development talks led by TPCB alumni. Students enjoyed various social activities from karaoke, hiking, boating, swimming and just exploring the beautiful Mohonk Mountain House property. Students appreciated hearing from alumni who offered useful insights about their respective careers in academia, administration and big pharma. Alumni guests included Disan Davis, PhD, (Program Manager, Science Outreach, Rockefeller University), Jarrod French, PhD, (Assistant Professor, Department of Biochemistry & Cell Biology, Stony Brook University), and Adam Levinson, PhD, (Research Scientist, Eli Lilly & Company).

The event was organized by TPCB students Ashley Chui (Bachovchin Lab, Sloan Kettering), Rachel Leicher (Liu Lab, Rockefeller), and Linamarie Miller (Klinge Lab, Rockefeller), and supported by the TPCB NIH T32 Chemistry-Biology Interface Training Grant.
July 2017
TPCB Student Fangyu Liu Determines Structures of Human Protein that Causes Cystic Fibrosis
 Cystic fibrosis arises from mutations in a single gene, CFTR (cystic fibrosis transmembrane conductance regulator), which encodes a chloride ion channel. Dysfunction of CFTR leads to accumulation of mucus in the lungs that can cause potentially fatal breathing problems and respiratory infections. In a recent Cell article, TPCB student Fangyu Liu and colleagues in the laboratory of Prof. Jue Chen at Rockefeller have reported the first molecular structure of the human CFTR protein using cryo-EM. This builds upon previous work in the lab determining the structure of CFTR from zebrafish, and reveals a previously unresolved helix in the R domain that that precludes channel opening. CFTR channel gating is strictly coupled to phosphorylation and ATP hydrolysis, and this study presented the channel in its dephosphorylated, ATP-free form. Electrophysiology experiments suggested that activation of the CFTR channel by R domain phosphorylation is enabled by infrequent, spontaneous disengagement of the R domain from the channel. Subsequently, in a second Cell paper, the team determined the structure of CFTR in its phosphorylated, ATP-bound conformation, in which major structural rearrangements lead to R domain disengagement and channel opening. Together, these reports provide new insights into the molecular mechanism of CFTR function and may ultimately inform the development of new therapeutics for cystic fibrosis to restore proper function of CFTR mutants. [Read more at: Rockefeller News | Read the articles in: Cell (23 Mar 2017) and Cell (27 Jul 2017)]
Cystic fibrosis arises from mutations in a single gene, CFTR (cystic fibrosis transmembrane conductance regulator), which encodes a chloride ion channel. Dysfunction of CFTR leads to accumulation of mucus in the lungs that can cause potentially fatal breathing problems and respiratory infections. In a recent Cell article, TPCB student Fangyu Liu and colleagues in the laboratory of Prof. Jue Chen at Rockefeller have reported the first molecular structure of the human CFTR protein using cryo-EM. This builds upon previous work in the lab determining the structure of CFTR from zebrafish, and reveals a previously unresolved helix in the R domain that that precludes channel opening. CFTR channel gating is strictly coupled to phosphorylation and ATP hydrolysis, and this study presented the channel in its dephosphorylated, ATP-free form. Electrophysiology experiments suggested that activation of the CFTR channel by R domain phosphorylation is enabled by infrequent, spontaneous disengagement of the R domain from the channel. Subsequently, in a second Cell paper, the team determined the structure of CFTR in its phosphorylated, ATP-bound conformation, in which major structural rearrangements lead to R domain disengagement and channel opening. Together, these reports provide new insights into the molecular mechanism of CFTR function and may ultimately inform the development of new therapeutics for cystic fibrosis to restore proper function of CFTR mutants. [Read more at: Rockefeller News | Read the articles in: Cell (23 Mar 2017) and Cell (27 Jul 2017)]
June 2017
TPCB Congratulates Our 2017 Graduates
Congratulations to the TPCB’s 2017 graduates Angelica Ferguson (Blanchard Lab, Weill Cornell), Adam Levinson (Danishefsky Lab, Sloan Kettering) and Carlos Rico (Sakmar Lab, Rockefeller). Our graduates received their degrees at the Weill Cornell Graduate School of Medical Sciences graduation ceremony on June 1, 2017 at Carnegie Hall. Students in Sloan Kettering labs were also recognized at the Gerstner Sloan Kettering Commencement Ceremony on May 18, 2017. Students in Rockefeller labs were recognized at The Rockefeller University Commencement Ceremony on June 15, 2017.
• Angelica Ferguson, PhD – “Functional Dynamics of the Human Ribosome Revealed by smFRET and Single-Particle cryoEM”
• Adam Levinson, PhD – “Part I: Total Synthesis of Aspeverin and Penicimutamide A; Part II: Total Chemical Synthesis and Folding of All-L and All-D KRas(G12V) and the Further Exploration of Isonitrile-Mediated Peptide Ligations”, Research Scientist, Eli Lilly & Company
• Carlos Rico, PhD – “Single-molecule Ligand Binding Studies on CCR5 by Fluorescence Cross-correlation Spectroscopy”, Postdoctoral Fellow, Prof. Thomas Sakmar, The Rockefeller University
Gregory Alushin, Joshua Levitz, Simon Scheuring, Thomas Walz, and Harel Weinstein Join the TPCB Faculty
TPCB recently welcomed two new junior and three new senior faculty members to the graduate program, bringing diverse research interests in the molecular mechanisms behind cytoskeletal rearrangement and membrane protein function:
• Prof. Gregory Alushin of Rockefeller studies the role of actin plasticity on the ability of cells to sense and respond to the mechanical properties of their surroundings. His lab uses molecular engineering to probe the forces involved in protein conformational changes. He received his PhD in 2012 from the University of California, Berkeley, under the guidance of Prof. Eva Nogales. After a brief postdoc with Dr. Clare Waterman at NHLBI, he received a prestigious NIH Director’s Early Independence Award and established his independent lab. Prof. Alushin joined TPCB and the Rockefeller faculty in January 2017. [Read more at: TPCB Faculty]
• Prof. Joshua Levitz of Weill Cornell uses high-resolution chemical and optical methods to study dynamic molecular processes involved in neurological signaling. His lab develops chemical optogenetic tools and single-molecule fluorescence-based assays to elucidate these fundamental processes. He carried out his PhD studies with Prof. Ehud Isacoff at the University of California, Berkeley, graduating in 2014. He then carried out postdoctoral training with Prof. Mu-ming Poo and Ehud Isacoff until 2016, when he joined TPCB and the Weill Cornell faculty. [Read more at: TPCB Faculty]
• Prof. Simon Scheuring of Weill Cornell is a leader in the use of atomic force microscopy to study the structure, dynamics, diffusion, interaction, mechanics, and supramolecular assembly of membrane proteins and other membrane constituents. He carried out his Diploma, PhD, and postdoctoral studies at the Biozentrum of the University of Basel in Switzerland. He was most recently a Senior Research Director in INSERM at Aix-Marseille Université in France. He joined TPCB and the Weill Cornell faculty in early 2017. [Read more at: TPCB Faculty]
• Prof. Thomas Walz of Rockefeller is interested in processes that involve biological membranes, particularly how lipids affect the structure and function of membrane proteins, and uses electron microscopy techniques to gain an atomic level understanding of these processes. He took his Diploma and PhD at the Biozentrum of the University of Basel in Switzerland, then went onto postdoctoral studies at the Krebs Institute at the University of Shefield in the UK. He was most recently a Professor and HHMI Investigator at Harvard Medical School in the Department of Cell Biology. He joined Rockefeller in 2015 and was appointed to the TPCB faculty in early 2017. [Read more at: TPCB Faculty]
• Prof. Harel Weinstein of Weill Cornell is a leader in computational chemistry and computational biophysics. His research uses computational modeling and simulation to understand the structural and dynamic mechanisms of cellular components. He completed his BSc, MSc, and DSc at Technion – Israel Institute of Technology and began his independent career at the Mount Sinai School of Medicine. He joined Weill Cornell as Professor and Chairman of the Department of Physiology and Biophysics in 2002 and was appointed to the TPCB faculty in early 2017. [Read more at: TPCB Faculty]
Daniel Bachovchin Receives Two Junior Faculty Awards
Prof. Daniel Bachovchin at Sloan Kettering has been named a 2017 Pew-Stewart Scholar in Cancer Research and has also been awarded a Stand Up To Cancer (SU2C) Innovative Research Grant. The Pew-Stewart Scholarship is a four-year award is funded by the Alexander and Margaret Stewart Trust and administered by the Pew Charitable Trusts to support promising early career scientists whose research will accelerate discovery and advance progress to a cure for cancer. The SU2C Innovative Research Grant program provides three years of support for cutting-edge cancer research by early-career scientists with novel, high-risk, high-impact ideas. Prof. Bachovchin’s research focuses on characterizing the functions of enzymes in normal and pathological processes, particularly the roles of serine proteases in cancer and immune system signaling. They use a variety of tools and approaches, including new chemical probes developed in the lab, mass spectrometry-based proteomics, activity-based protein profiling, and genome engineering (CRISPR). He is the first TPCB faculty member to receive each of these honors. [Read more at: Pew-Stewart Scholars | Stand Up To Cancer]
May 2017
TPCB Student Adam Levinson Targets “Undruggable” Target
The oncoprotein KRas acts as a switch that drives cellular growth and survival. Mutations in KRas are linked to nearly 30% of all cancers, making it an attractive target for cancer therapy. However, KRas has long been considered an “undruggable” target, in large part because it binds extremely tightly to its natural substrate GTP. As a result, traditional drug discovery approaches have failed to discover effective KRas inhibitors. In a new paper in J. Am. Chem. Soc., recent TPCB graduate Adam Levinson and colleagues in the lab of Prof. Samuel Danishefsky at Sloan Kettering and Prof. Gregory Verdine at Harvard report an innovative approach to tackling this challenge, leveraging fundamental concepts in stereochemistry. Using yeast display, massive libraries of L-peptides are presented on the cell surface and can be selected for binding to the target of interest, enabling directed evolution to identify potent ligands. However, because L-peptides are readily degraded by proteases, they generally do not make effective drugs without modifications to evade this process. Thus, in the mirror-image approach, the D-enantiomer of the target is instead used in the screen. The L-peptide ligands that are discovered are then synthesized chemically as their D-peptide enantiomers, which analogously bind the original L-protein target. Importantly, these D-peptide ligands are not subject to proteolysis, and serve as attractive starting points for further drug development efforts. To enable this approach, the team executed the total chemical synthesis of the D-enantiomer of entire KRas protein in the lab, using native chemical ligation as well as related peptide ligation strategies developed in the Danishefsky lab over the last decade. They subsequently showed that this mirror-image protein folded properly and bound to the corresponding unnatural enantiomer of a non-hydrolyzable GTP analogue. This tour de force in chemical synthesis now sets the stage for screening efforts to discover novel KRas inhibitors based on D-peptides. [Read more at: MSK News | Read the article in: J. Am. Chem. Soc.]
NYAS 2017 Chemical Biology Symposium Brings Together NYC Scientists
The New York Academy of Sciences (NYAS) held its annual year-end Chemical Biology Symposium on May 24, 2017, in partnership with TPCB. This annual meeting brings together chemical biology researchers from universities and pharmaceutical companies across the tristate area and is a highlight of the NYAS Chemical Biology Discussion Group’s annual symposium series. This year’s event featured two keynote speakers, TPCB faculty member Prof. Sean Brady of Rockefeller as well as Prof. Dirk Trauner, who recently relocated his lab to NYU. Prof. Brady presented his lab’s recent discovery of humimycin as a novel antibiotic active against methicillin-resistant Staphylococcus aureus infections. The compound was discovered using bioinformatic analysis of genomes from human gut bacteria. Prof. Trauner shared his lab’s efforts to develop photopharmacology tools to provide precise control over protein function, with potential applications in vision restoration. Students and postdocs from TPCB labs as well as other labs across the NYC area presented posters on their research. Several were selected for oral presentations, including one by Dr. Corinne Foley from the lab of TPCB faculty member Prof. Derek Tan, who discussed the discovery of novel antitrypanosomal alkaloids from a diversity-oriented synthesis library. Several poster prizes were also awarded, including one to Dr. Maria Chiriac, also from the Tan lab, who described her work on developing novel macrocyclic inhibitors of bacterial adenylation enzymes. The event was co-organized by TPCB faculty member Prof. Howard Hang of Rockefeller. [Read more at: NYAS]
Morgan Huse Receives Boyer Young Investigator Award
Prof. Morgan Huse at Sloan Kettering has been honored with the Louise and Alston Boyer Young Investigator Award for Excellence in Basic Research, which recognizes outstanding junior faculty at MSK. Prof. Huse’s lab combines imaging technology with synthetic chemistry and materials science to understand how immune cells communicate. He joins several other TPCB faculty members who received this honor in recent years: Christopher Lima (2006), Stephen Long (2016), Derek Tan and (2010). [Read more at: MSK News]
April 2017
Christopher Lima Elected Fellow of the American Academy of Arts and Sciences
Prof. Christopher Lima at Sloan Kettering has been named a Fellow of the American Academy of Arts and Sciences. Election to the Academy is one of the most significant honors one can receive and recognizes leadership in addressing critical challenges facing society. Prof. Lima’s lab uses structural, biochemical, and functional approaches to study molecules involved in RNA processing and protein modification. His lab investigates on how ubiquitin and the related protein SUMO are attached to other proteins to modify their function or fate. His lab also focuses on systems that contribute to RNA stability and degradation. He is the seventh TPCB faculty member to named a Fellow, joining C. David Allis (2001), Lewis Cantley (1999), Samuel Danishefsky (1984), Dinshaw Patel (2014), Nikola Pavletich (2014), and Stewart Shuman (2015). [Read more at: AAAS]
March 2017
TPCB Students Mehtap Isik and Rafal Wiewora Awarded Prestigious Fellowships
Two TPCB students have been awarded prestigious fellowships to support their graduate research!
Mehtap Isik has received the 2017 Hutchinson Fellowship from the Sloan Kettering Division of the Weill Cornell Graduate School of Medical Sciences. The award is named after Dr. Dorris J. Hutchinson, who was a microbiologist in the Sloan Kettering Institute for nearly 40 years studying new treatments for tuberculosis and leukemia. Dr. Hutschinson was also instrumental in establishing the Sloan Kettering Division of the Weill Cornell Graduate School and this award supports outstanding students pursuing their thesis research in this program at Sloan Kettering. Mehtap’s project focuses on the development of model protein–ligand systems to advance the field of predictive quantitative computational modeling for drug discovery. Her project uses a combination of robotic wetlab experiments and advanced GPU computing. She carried out her undergraduate training at Boğaziçi University in Turkey. [Read more at: MSK News]
Rafal Wiewiora has been awarded a Horizon Award by the Department of Defense’s Peer Reviewed Cancer Research Program. This two-year grant supports exceptional junior-level scientists pursuing research in cancer with the guidance of a faculty mentor. Rafal’s project investigates the conformational heterogeneity of histone methyltransferases using molecular dynamics simulations on the Folding@home platform, a worldwide distributed computing project where hundreds of thousands of people around the world contribute their computing power toward the understanding of cancer targets. These models of conformational dynamics will be vital in aiding the in silico design of selective chemical probes to understand the roles that these methyltransferases play in cancer, as well as to develop new strategies to inhibit them. A native of Poland, Rafal received his MChem degree from the University of Oxford. [Read more at: DOD]
Both Mehtap and Rafal are third-year TPCB students working in the lab of Prof. John Chodera at Sloan Kettering.
January 2017
Gregory Alushin Receives Presidental Early Career Award for Scientists and Engineers
Prof. Gregory Alushin at Rockefeller has a Presidential Early Career Award for Scientists and Engineers. Established in 1996, this award represents the highest honor given by the U.S. government to science and engineering professionals in the early stage of their careers. Prof. Alushin’s lab studies how cells use their structural filaments to sense and respond to mechanical forces. Using cryo-electron microscopy along with biochemical and biophysical approaches, his lab aims to understand how changes to actin—a predominant class of protein filaments—are linked to alterations in gene expression in development and disease. He is the first TPCB faculty member to receive this honor. [Read more at: Rockefeller News | Office of the White House Press Secretary | NIH]
December 2016
TPCB Student Malik Chaker-Margot Solves Structure of Key Intermediate in Ribosome Assembly
The assembly of eukaryotic ribosomes, the cell’s protein-synthesizing factories, is a complicated process that is not yet fully understood. This is partly because the enormous size and complexity of the eukaryotic ribosomal machinery complicate efforts to obtain atomic-level structural information for key intermediates. Now, in a breakthrough study reported in Science, TPCB student Malik Chaker-Margot and colleagues in the lab of Prof. Sebastian Klinge at Rockefeller, report the 5.1 Å cryo-electron microscopy (cryo-EM) structure of an important intermediate in ribosome assembly, called the small subunit (SSU) processome. The yeast SSU processome is a giant ribonucleoprotein particle, composed of approximately 70 proteins and several RNAs, that is involved in the assembly of the small eukaryotic ribosomal subunit. By examining the cryo-EM structure, the team gained important insights that led them to develop a new model for SSU processome assembly, in which an RNA leader sequence participates during the maturation of the small ribosomal subunit. [Read more at: Rockefeller News | Read the article in: Science]
November 2016
TPCB Student John Zinder Uses Click Chemistry to Capture Nuclear RNA Exosome Structure
RNA decay is an essential cellular process that is catalyzed in part by the RNA exosome, a multi-subunit exoribonuclease complex that degrades RNA in the 3´-to-5´ direction. In a recent paper in Molecular Cell, TPCB student John Zinder and colleagues in the lab of Prof. Christopher Lima at Sloan Kettering describe the first high-resolution crystal structure of an 11-subunit nuclear RNA exosome. The team leveraged alkyne–azide click chemistry to synthesize an engineered RNA having two 3´-ends designed to bind concurrently to two exoribonuclease active sites, one in Rrp6 and one in Rrp44. The crystal structure offers valuable information on the way the RNA substrate is routed through the nuclear exosome complex. [Read the article in: Mol. Cell]
Yueming Li Elected Fellow of the American Association for the Advancement of Science
TPCB faculty member Prof. Yueming Li at Sloan Kettering has been elected a Fellow of the American Association for the Advancement of Science. This honorific recognizes AAAS members whose efforts on behalf of the advancement of science or its applications in service to society have distinguished them among their peers and colleagues. Prof. Li is a leader in studying the function, regulation, and inhibition of intramembrane proteases, the most prominent of which is γ-secretase, the enzyme implicated in Alzheimer’s disease. He began his career at Merck Research Laboratories in 1997 where he led the team that discovered presenillin as the catalytic subunit of γ-secretase. He joined the Sloan Kettering and the TPCB faculty in 2002, where his lab has continued to elucidate the components of the γ-secretase complex using biochemical and chemical approaches, reconstituted γ-secretase using recombinant proteins, and studied its functional roles in neurodegenerative disorders and cancer. [Read more at: AAAS]
October 2016
Sebastian Klinge Receives $1.5M NIH New Innovator Award
Prof. Sebastian Klinge at Rockefeller has been awarded a prestigious Director’s New Innovator grant by the NIH. This $1.5 million, five-year grant will support his lab’s research into assembly of the eukaryotic ribosome. Ribosomes are essential molecular machines that catalyze protein synthesis in cells. The lab’s research focuses on elucidating both the structural anatomy of the ribosome assembly machinery as well as the underlying biochemical transformations involved in the assembly process. Prof. Klinge joined the Rockefeller faculty as an Assistant Professor in 2013 and joined the TPCB faculty shortly thereafter. He is the seventh TPCB faculty member to have received NIH Director’s New Innovator Award since the program’s inception in 2007. [Read more at: Rockefeller News | NIH]
September 2016
TPCB Student Zhen Chen Identify Chemical Probes of Eukaryotic Ribosome Biogenesis
Ribosomes, the complex protein-building machinery of the cell, are assembled at a rate of about 2,000 per minute. However, a lack of suitable chemical probes has made it difficult to decipher the mechanisms underlying this dynamic and essential process in eukaryotes. In a new Cell paper, TPCB student Zhen Chen and colleagues in the lab of Prof. Tarun Kapoor at Rockefeller, describe novel small-molecule probes of ribosome assembly. The Kapoor lab employed chemical and genetic approaches to identify the ribozinoindoles (Rbins) as potent, reversible, cell-permeable inhibitors of eukaryotic ribosome biogenesis. Chemical synthetic lethal screening of a >10,000 compound library in fission yeast revealed ribozinoindole-1 (Rbin-1) to be selectively toxic to cells possessing point mutations in mdn1, an essential gene required for eukaryotic ribosome biogenesis. Several different point mutations in mdn1 were found to confer chemotype-specific resistance to Rbin-1 in cells. Biochemical studies revealed direct inhibition of the ATPase activity of Mdn1 by Rbin-1. Furthermore, the same mutation that protected cells from Rbin-1 also suppressed inhibition of enzyme activity in vitro. In cell-based studies, the group used Rbins to acutely inhibit or activate Midasin function and discovered that Midasin plays multiple roles in assembling precursors of the 60S subunit. In addition to their value as chemical probes of ribosome biogenesis, the Rbins could form the basis for new drugs to treat fungal infections through a novel mechanism of action. [Read more at: Rockefeller News | Read the article in: Cell]
Bachovchin Lab Identifies New Mechanistic Pathway in Cancer Immunotherapy
Cancer immunotherapies, drugs that induce the immune system to attack tumor cells, show great promise in the clinic. However, limitations associated with existing therapies create the impetus to explore new immunotherapeutic mechanisms. Reporting in Nature Chemical Biology, Prof. Daniel Bachovchin and his group at Sloan Kettering, which includes TPCB students Darren Johnson, Eun Bin Go, and Ashley Chui, describe a small molecule that stimulates an immune response against cancer through an unusual mechanism called pyroptosis. Pyroptosis is a form of immune cell death wherein the cell swells, bursts, and releases pro-inflammatory signaling molecules into the environment, which then triggers an immune response. The small molecule Val-boroPro, originally designed to inhibit the serine protease DPP4, is known to induce persistent tumor immunity in mice. However, the mechanism of this immunoresponse had remained a mystery. The Bachovchin lab used activity-based protein profiling to test Val-boroPro for inhibition of ≈100 recombinant serine hydrolases in biochemical assays. They discovered that the molecule potently inhibits DPP8 and DPP9 and that inhibition of these two serine proteases leads to direct activation of pro-caspase-1. The activated pro-protein fails to self-cleave to mature caspase-1 and instead cleaves and activates gasdermin D, leading to induction of pyroptosis in two types of immune cells. Val-boroPro is the first small molecule known to induce an immunoresponse through pyroptosis. Further studies into this immune response mechanism could lead to a new class of cancer immunotherapeutics. [Read more at: MSK News | Read the article in: Nat. Chem. Biol.]
Luciano Marraffini Named HHMI-Simons Faculty Scholar
TPCB faculty member Prof. Luciano Marraffini at Rockefeller has been named a Howard Hughes Medical Institute Faculty Scholar. This new program supports early-career scientists who have great potential to make unique contributions to their fields and was established recently by HHMI, The Simons Foundation, and The Bill & Melinda Gates Foundation. Prof. Marraffini’s lab studies the fundamental mechanisms behind CRISPR-based immunity in bacteria and other microorganisms. The system enables the bacteria to remember the DNA sequences of invading phages, so they can cleave viral DNA upon subsequent infections. In 2008, while a postdoc at Northwestern University, Dr. Marraffini demonstrated that CRISPR immunity targets the DNA of the invader, rather than the RNA as previously thought. More recently, his lab at Rockefeller has gained important insights into the roles of the enzyme Cas9 in acquisition and implementation of this immunity, as well as our understanding of how bacteria distinguish between harmful and beneficial viruses. Prof. Marraffini joined the Rockefeller faculty in 2010 and was promoted to Associate Professor earlier this year. [Read more at: Rockefeller News | HHMI]
Yael David, Richard Hite, and Pengbo Zhou Hoin the TPCB Faculty
TPCB recently welcomed three new faculty members to the program, bringing diverse research interests in chemical epigenetics, ion channel structure, and protein degradation.
• Prof. Yael David of Memorial Sloan Kettering is using a novel semisynthetic approach to generate histone proteins bearing specific post-translational modifications that impact chromatin remodeling. Her lab is interested in probing the roles and regulation of these epigenetic modifications in cancer biology and neuroscience. She received her PhD in 2011 from The Weizmann Institute under the guidance of Prof. Ami Navon. From 2011–2016, she carried out postdoctoral training in Prof. Tom Muir’s lab at Princeton. Prof. David joined TPCB and the MSK faculty in September 2016. [Read more at: TPCB Faculty]
• Prof. Richard Hite of Memorial Sloan Kettering is interested in studying the structures and functions of lysosomal cation channels that regulate lysosomal trafficking, fusion, and catabolite efflux. His lab leverages expertise in cryo-electron microscopy, X-ray crystallography, biochemistry, and biophysics to elucidate mechanisms of gating and selectivity in these channels. He carried out his PhD studies with Prof. Thomas Walz at Harvard, graduating in 2010. He then joined Prof. Rod MacKinnon’s lab at Rockefeller for his postdoctoral training from 2011–2016. Prof. Hite joined TPCB and the MSK faculty in September 2016. [Read more at: TPCB Faculty]
• Prof. Pengbo Zhou of Weill Cornell studies the molecular mechanisms by which ubiquitin-dependent proteolysis operates under physiological conditions and how dysregulation of the ubiqutin-proteasome pathway contributes to cancer and neurological diseases. His lab uses a combination of chemical, biochemical, cell and molecular biological, and mouse genetic approaches to study the Cullin family of ubiquitin ligases as potential anticancer drug targets. Prof. Zhou has been a member of the Weill Cornell faculty since 1999. [Read more: TPCB Faculty]
August 2016
Chemical Biology Shines at the 12th Annual Tri-Institutional Symposium
TPCB hosted the 12th Annual Chemical Biology Symposium on August 17, 2016 at MSK. The student-organized event was attended by over 180 undergraduates, graduate students, postdocs, staff scientists, and faculty, and included seminars by emminent faculty keynote speakers and TPCB students, and a vibrant poster session highlighting chemical biology research across the Tri-Institutional Research Program. Prof. Matthew Shair from Harvard kicked off the day with an enthralling presentation of his lab’s work using small-molecule inhibitors based on the natural product cortistatin to study the role of Mediator complex kinases in leukemia. TPCB student Adam Levinson, from the lab of Prof. Samuel Danishefsky, then presented his impressive total synthesis of the D-enantiomer of the K-Ras oncoprotein for inhibitor discovery using mirror image phage display. Prof. Ming Hammond from Berkeley shared her lab’s exciting research on riboswitch-based approaches to studying bacterial and immune signaling. TPCB student Malik Chaker-Margot closed out the morning session with his seminal work illuminating stage-specific assembly of processome proteins involved in maturation of the eukaryotic ribosome.
Symposium participants then enjoyed lunch and a poster session featuring the latest research advances from chemical biology students, staff, and faculty from labs across the entire Tri-Institutional Research Program. Poster awards went to Zhen Chen (Kapoor Lab, RU), Alexis Jaramillo (Darst Lab, RU), John Zinder (Lima Lab, MSK), and Matthew Streeter (David Spiegel Lab, Yale University). In the afternoon, TPCB faculty member Prof. Scott Blanchard of Weill Cornell presented his lab’s exciting new work on single-molecule imaging of integral membrane proteins. TPCB student Rudolf Píša followed by sharing his work in the lab of Prof. Tarun Kapoor at Rockefeller to develop selective small-molecule inhibitors of AAA+ enzymes. Finally, Prof. Laura Kiessling from the University of Wisconsin-Madison spoke about her lab’s intriguing recent discovery of selective microbial carbohydrate binding by the human intelectin-1 protein. The day concluded with a reception at which students, postdocs, and faculty continued to discuss the latest exciting advances in chemical biology. The event was co-chaired by TPCB students Michaelyn Lux (Tan Lab, MSK) and Jacob Litke (Jaffrey Lab, Weill Cornell), with support from TPCB faculty mentor Prof. Daniel Heller (MSK), and received generous promotional and poster prize support from ACS Chemical Biology, Nature Chemical Biology, the Royal Society of Chemistry, and the New York Academy of Sciences.
Lewis Cantley and Neal Rosen Named NCI Outstanding Investigators
TPCB faculty members Prof. Lewis Cantley at Weill Cornell and Prof. Neal Rosen at Sloan Kettering have received Outstanding Investigator Awards from the National Cancer Institute. These prestigious grants were established in 2015 to support to accomplished investigators with outstanding records of cancer research productivity who propose to conduct exceptional research. The $4.2M award provides long-term support over a seven-year period and is intended to allow investigators the opportunity to take greater risks and be more adventurous in their research. [Read more at: NCI]
Prof. Cantley’s research focuses on studying the biochemical pathways by which oncogenes mediate changes in cellular metabolism, growth, and transformation. His lab discovered the phosphoinositide 3-kinase (PI3K) enzymes in 1984 and has found that the PI3K pathway controls cell growth primarily through regulation of cellular metabolism. His lab is now investigating drug combinations with PI3K inhibitors that are likely to be more effective than single inhibitors. They are also developing small-molecule inhibitors of phosphatidylinositol-5-phosphate 4-kinases to investigate the cellular functions of these enzymes and their potential as new therapeutic targets. Prof. Cantley began his career at Harvard and Tufts and joined Weill Cornell and the TPCB faculty in 2012. [Read more at: TPCB Faculty]
Prof. Rosen’s lab studies signal transduction pathways that cause dysregulation of growth and inhibition of apoptosis in advanced human cancers. His current research focuses on understanding the roles of feedback inhibition by oncoproteins in both physiologic and oncogenic signaling. This work has critical implications for the use of small molecule anticancer drugs that inhibit oncoprotein-activated signaling but concurrently reactivate physiologic signaling pathways and promises to enable the development of rational combination therapies that target these reactivated pathways. Prof. Rosen began his career at the NCI and joined the Sloan Kettering faculty in 1992. He has been a TPCB faculty member since the program’s inception in 2001. [Read more at: TPCB Faculty]
July 2016
TPCB Student Hala Iqbal Develops New Platform for Metagenomic Natural Product Discovery
Bacteria have historically served as a rich source of novel natural products with diverse biological activities, including many clinically used drugs. However, the vast majority of bacteria cannot be cultured in the lab, so identifying natural products from these bacteria requires development of methods for heterologous expression of the underlying biosynthetic genes from metagenomic sources such as soil samples. To address this problem, recent TPCB graduate Hala Iqbal, PhD and Prof. Sean Brady at Rockefeller have screened a collection of 39 strains of Streptomyces bacteria for their ability to support heterologous expression of natural product biosynthetic pathways. Streptomyces are well known to produce diverse natural products native to the individual strains, but have thus far been of limited utility for heterologous expression. They identified Streptomyces albus as the most effective heterologous host, then used it to screen over one million metagenomic DNA cosmids. This led to the discovery of a novel tricyclic natural product, which they named metatricycloene. This new heterologous expression platform should now facilitate discovery of other natural products with novel structures and biological activities. [Read the article in: J. Am. Chem. Soc.]
TPCB Graduate Amrita Hazra Launches Her Independent Lab at IISER-Pune
Congratulations to TPCB alumna Amrita Hazra, PhD who recently joined the Indian Institute of Science Education and Research (IISER), Pune as an Assistant Professor in Chemistry & Biology. Prof. Hazra’s lab investigates the genetic pathways and molecular and enzymatic mechanisms involved in microbial metabolism. [Read more at: IISER]
She joins four other TPCB alumni in tenure-track faculty positions:
• Jarrod French, PhD (Assistant Professor, Department of Biochemistry & Cell Biology, Stony Brook University) studies the structure, function, and control of enzymes and enzyme complexes involved in cellular metabolism. [Read more at: Stony Brook U.]
• Niroshika Keppetipola, PhD (Assistant Professor, Department of Chemistry & Biochemistry, California State University Fullerton) explores how posttranslational modifications alter the activity of splicing factors and, in turn, cellular gene expression. [Read more at: CSU Fullerton]
• Jayakrishnan Nandakumar, PhD (Assistant Professor, Department of Molecular, Cellular, & Developmental Biology, University of Michigan) investigates how telomeres and telomerase participate in cancer, stem cell fitness, aging, and genome stability. [Read more at: U. Michigan]
• Brian Zoltowski, PhD (Assistant Professor, Department of Chemistry, Southern Methodist University) studies the molecular mechanisms of blue-light photoreceptors. [Read more at: SMU]
TPCB’s over 60 PhD alumni have used their training in chemical biology to pursue research careers in universities, major pharmaceutical companies, biotechnology firms, research institutes, and government labs, and to take up leading positions in science policy, outreach, education, consulting, and patent law. [Read more at: TPCB Alumni]
June 2016
TPCB Congratulates Our 2016 Graduates
Congratulations to the TPCB’s 2016 graduates Debjani Chakraborty (Brady Lab, Rockefeller), Stefanie Gerstberger (Tuschl Lab, Rockefeller), Han Guo (Luo Lab, Sloan Kettering), David Iaea (Maxfield Lab, Weill Cornell), Hala Iqbal (Brady Lab, Rockefeller), Thinh Nguyen Duc (Huse Lab, Sloan Kettering), Xiaoqiu Yuan (Hang Lab, Rockefeller), Qinsi Zheng (Blanchard Lab, Weill Cornell). Our graduates received their degrees at the Weill Cornell Graduate School of Medical Sciences graduation ceremony on May 25, 2016 at Carnegie Hall. Students in Sloan Kettering labs were also recognized at the Gerstner Sloan Kettering Commencement Ceremony on May 19, 2016. Students in Rockefeller labs were recognized at The Rockefeller University Commencement Ceremony on June 9, 2016.
• Debjani Chakraborty, PhD – “Studies towards the Discovery of Novel Natural Products through Functional Analysis of Environmental-DNA Derived Type II Polyketide Synthases”, Postdoctoral Fellow, Prof. Kamlesh Yadav, Mount Sinai
• Stefanie Gerstberger, PhD – “Genome-wide Targeted Approaches to the Study of PTGR and Identification and Characterization of NEF-sp in the 3’ End Maturation of 28S rRNA”, Medical Student, PhD to MD Program, Columbia University
• Han Guo, PhD – “Bioorthogonal Substrate Profiling and Functional Studies of Arginine Methylation”, Business Development Manager, Betta Pharmaceuticals Co., Ltd.
• David Iaea, PhD – “Structure & Function Studies of the Sterol Transport Protein, STARD4”, Associate Scientist, Genentech
• Hala Iqbal, PhD – “Mining Natural Products from Uncultured Bacteria: Functional Screening of Soil Metagenomic Libraries for Antibacterial and Colored Compounds”, Postdoctoral Fellow, Prof. Sean Brady, The Rockefeller University
• Thinh Nguyen Duc, PhD – “Photochemical Approaches to Control Cell Surface Receptors Activation with Spatiotemporal Specificity”, Senior Life Sciences Specialist, L.E.K. Consulting
• Xiaoqiu Yuan, PhD – “Chemical Tools for Exploring IFITM3 S-palmitoylation and Mechanism”, Equity Research Associate, Credit Suisse
• Qinsi Zheng, PhD – ““Self-healing” Organic Fluorophores for Single-molecule Microscopy and Spectroscopy”, Postdoctoral Fellow, Professor Robert Singer, Janelia Research Campus
May 2016
David Scheinberg Wins Award for Excellence in Teaching and Mentoring
TPCB faculty member Prof. David A. Scheinberg at Sloan Kettering has been awarded the 2016 Excellence in Teaching and Mentoring Award by the Weill Cornell Graduate School, in recognition of his exceptional efforts in mentoring both graduate students and junior faculty during his 26 years on the faculty. Since joining the WCGS faculty in 1990, Prof. Scheinberg has trained 24 PhD graduate students and 34 postdoctoral fellows, many of whom have gone onto successful careers in academia and industry. Prof. Scheinberg was nominated by former trainees who cited his steadfast support, scientific creativity, and collaborative spirit as critical to their success in tackling challenging new problems in chemical biology and biomedical research. This award also recognizes Prof. Scheinberg’s efforts over the last 15 years to recruit and mentor junior faculty, including current TPCB faculty members Prof. Derek Tan, Prof. Yueming Li, Prof. Gabriela Chiosis, Prof. Minkui Luo, Prof. Alex Kentsis, and Prof. Daniel Heller. In this role, he was instrumental in building the chemical biology research program at MSK.
Prof. Scheinberg received his AB from Cornell University and completed his MD-PhD training at Johns Hopkins. He then carried out his internship and residency at Weill Cornell, then continued onto a fellowship in oncology-pharmacology at MSK. He joined the faculty in 1989 was promoted through the ranks to Member at MSK and Professor at Weill Cornell in 1999. He served as Chairman of the MSK Molecular Pharmacology & Chemistry Program from 2001–2015 and is currently Chairman of the Molecular Pharmacology Program. Prof. Scheinberg has also co-chaired the WCGS Pharmacology graduate program since 1990 with Prof. Lorraine Gudas of Weill Cornell, also a TPCB faculty member. [Read more at: MSK News]
Kentsis Lab Research Program Featured on Humans of New York
TPCB faculty member Prof. Alex Kentsis and his lab’s research program has been featured by Humans of New York for his important work in pediatric cancer research. Prof. Kentsis notes that advances in understanding the molecular basis for cancers are leading to new opportunities for therapy. His research program in chemical biology uses next-generation proteomic approaches to investigate the mechanisms by which genomes and proteomes of tumors are disorganized and to use this information to develop rational cancer therapies. Humans of New York was created by photojournalist Brandon Stanton in 2010 to feature photographs and interviews collected on the streets of New York City. Prof. Kentsis’s lab was featured as part of a Humans of New York fundraising campaign for pediatric cancer research at MSK that raised over $3.8 million in one month, from over 100,000 individuals. [Read more at: Humans of NY]
NYAS 2016 Chemical Biology Symposium Focuses on Chemical Approches to Epigenetics
TPCB partnered with the New York Academy of Sciences to present the annual year-end NYAS Chemical Biology Symposium on May 25, 2016. The meeting is part of the NYAS Chemical Biology Discussion Group series, which brings together numerous chemical biology laboratories from the greater NYC area to promote collaborations and exchanges of ideas. This year’s symposium featured two keynote speakers Prof. Philip A. Cole of Johns Hopkins and Dr. Kenneth W. Duncan of Epizyme. Prof. Cole spoke about his work using chemical approaches to study acetylation and methylation of protein lysine residues. Dr. Duncan presented Epizyme’s work to develop novel small-molecule anticancer drugs that target methyltransferase enzymes. Numerous students and postdoctoral fellows from participating NYC-area institutions presented talks and posters, including several from TPCB labs. [Read more at: NYAS]
January 2016
Lewis Cantley Awarded 2016 Wolf Prize in Medicine for Discovery of PI3K
TPCB faculty member Prof. Lewis Cantley of Weill Cornell has been awarded the 2016 Wolf Prize in Medicine by the Wolf Foundation. Prof. Cantley is recognized for his groundbreaking discovery of the enzyme phosphoinositide-3-kinase (PI3K) and the signaling pathway that it controls. PI3K is aberrantly activated in various cancers and the first drug targeting this enzyme, idelalisib, was recently approved for the treatment of chronic lymphocytic leukemia. Prof. Cantley will share the award with Prof. C. Ronald Kahn of Harvard Medical School, with whom he has collaborated to elucidate links between insulin, PI3K, and diabetes. Prof. Cantley is also the Director of the Sandra and Edward Meyer Cancer Center. [Read more at: Weill Cornell News]
December 2015
Angelica Ferguson and Scott Blanchard Probe Structural Dynamics in Ribosomal Translation
Protein translation requires carefully orchestrated motions of tRNAs through the ribosome active site. To probe these events in real time, TPCB student Angelica Ferguson and TPCB faculty member Prof. Scott Blanchard at Weill Cornell have used state-of-the-art, single-molecule fluorescence imaging approaches to monitor conformational transitions of tRNAs during protein translation. By labeling three individual tRNAs with distinct fluorescent dyes, they used FRET (Förster resonance energy transfer) imaging to measure distance changes between the tRNAs during single-turnover and processive translation reactions. This revealed hidden sub-states of tRNA motions that are stabilized by ribosome-targeting small-molecule antibiotics, providing new insights into the molecular mechanisms of action of these compounds. They also discovered long-range allosteric interactions between spatially separated tRNA binding sites, shedding new light on how conformational dynamics can regulate processive translation. This platform will be invaluable for future studies into the impacts of various protein factors and small-molecule inhibitors on ribosome dynamics. [Read the article in: Mol. Cell]
November 2015
Gabriela Chiosis, Tarun Kapoor, and Derek Tan Quoted as Voices of Chemical Biology
As part of its 10th anniversary, Nature Chemical Biology asked leading chemical biologists for their views on a variety of issues pertinent to the field. Three TPCB faculty members were quoted during the series, which appeared throughout 2015. Prof. Gabriela Chiosis of Sloan Kettering offered her definition of chemical biology in the June issue, her views on the most important contributions of the field in the July issue, and her advice to young chemical biologists in the November issue. Prof. Tarun Kapoor of Rockefeller and Prof. Derek Tan of Sloan Kettering both shared their thoughts on what they valued most about being part of the chemical biology community in the October issue. Prof. Tan also cited effective communication between chemists and biologists as one of the most significant challenges in the field in the August issue. [Read the articles in Nat. Chem. Biol.: June | July | August | October | November]
October 2015
Malik Chaker-Margot and Sebastian Klinge Probe Ribosome Assembly Machinery
The ribosome is a complex molecular machine that is comprised of 79 proteins and four rRNA components in eukaryotes. Ribosome assembly is an elaborate process that occurs while the pre-rRNA precursor is being transcribed. The earliest part of this process is carefully coordinated by the ‘small-subunit processome’, a complex of some 70 proteins, many of whose functions are unknown. To determine the temporal order in which small-subunit processome components associate with pre-rRNA at various stages of its transcription, TPCB student Malik Chaker-Margot and TPCB faculty member Prof. Sebastian Klinge at Rockefeller have developed a novel chemical biology approach involving affinity purification of complexes formed on truncated pre-rRNAs of various lengths. They then used mass spectrometry to identify the proteins associated with each truncated pre-rRNA, leading them to propose a revised model for early eukaryotic ribosome assembly involving consecutive recruitment of small-subunit processome components on pre-rRNA domains. This work provides fundamental insights into ribosome assembly and may one day lead to new therapies for rare human disorders that arise from defects in this process. [Read more at: Rockefeller News | Read the article in: Nat. Struct. Mol. Biol. | Read highlight in: Nat. Struct. Mol. Biol.]
Nikola Pavletich Elected to National Academy of Medicine
TPCB faculty member Prof. Nikola Pavletich at Sloan Kettering has been elected to the National Academy of Medicine, formerly the Institute of Medicine of the National Academies. Membership in the Academy is one of the highest honors in the fields of health and medicine and recognizes individuals for outstanding professional achievement and commitment to service. Prof. Pavletich is a leading structural biologist whose work focuses on X-ray crystallographic and biochemical studies of proteins that play key roles in pathways altered in cancer, including the DNA damage response and cell cycle. He joins TCPB faculty member Prof. Lewis Cantley of Weill Cornell, who was elected to the Academy last year. Prof. Pavletich is also Chairman of the Structural Biology Program at Sloan Kettering and has been a Howard Hughes Medical Institute Investigator since 1997. [Read more at: National Academy of Medicine | The Cancer Letter | The ASCO Post]
September 2015
Kapoor and Chait Labs Shed Light on Fundamental Process of DNA Repair
TPCB faculty members Prof. Tarun Kapoor and Prof. Brian Chait, both at Rockefeller, have collaborated to gain new molecular insights into the recruitment of DNA repair proteins to damaged sites on chromosomes. Using a quantitative chemical proteomic method that the team first reported in 2012, involving photochemical cross-linking, they discovered that a DNA repair protein known as 53BP1 interacts with phosphorylated histone H2AX, a post-translational modification that occurs in response to DNA damage. Such findings are vital to our understanding of disease etiology and may aid in the development of drugs that target DNA repair. [Read more at: Rockefeller News | Read the article in: Nat. Chem. Biol.]
Derek Tan Featured in CUNY “Science Forward” Video Series
Prof. Derek Tan, TPCB Director and faculty member at Sloan Kettering, is one of the scientists featured in the “Science Forward” video series produced by the Macaulay Honors College at the City University of New York (CUNY). In the video on “Drug Development and Discovery”, Prof. Tan talks about the wide range of skills and approaches chemical biologists use to design and synthesize new potential drugs. Viewers can catch a glimpse of Prof. Tan’s lab as well as TPCB student Michaelyn Lux in action. In the final video series, “Scientific Uncertainty”, Prof. Tan is quoted on the exciting role of uncertainty in his research. [View the videos]
August 2015
TPCB Symposium Highlights Chemical Biology Research Across Tri-Institutional Program
TPCB hosted the 11th Annual Chemical Biology Symposium on August 28, 2015 at MSK, bringing together chemical biology students and faculty from across the Tri-Institutional Research Program and other institutions in New York City. The program featured four distinguished keynote speakers: Prof. Catherine L. Drennan from MIT, Prof. Sarah E. Reisman from Caltech, Prof. Peter G. Schultz from Scripps, and TPCB faculty member Prof. Morgan Huse from MSK. TPCB students Angelica Ferguson (Blanchard Lab, Weill Cornell), Adam Trotta (Danishefsky Lab, MSK), and Stefanie Gerstberger (Tuschl Lab, Rockefeller) also gave oral presentations on their thesis research. The event featured posters from over 30 students and postdocs from across the Tri-Institutional Research Program, with Best Poster Awards given to John Zinder (Lima Lab, MSK), David Iaea (Maxfield Lab), Weill Cornell, and Alexis Jaramillo (Darst Lab, RU).
July 2015
Daniel Bachovchin, Steven Gross, Xuejun Jiang, and Shixin Liu Join the TPCB Faculty
TPCB recently welcomed four new faculty members to the program, bringing diverse research interests in serine proteases, metabolomics, cell death, and gene regulation.
• Prof. Daniel Bachovchin of Memorial Sloan Kettering is interested in characterizing the roles that serine proteases play in cancer and immune system signaling using activity-based protein profiling, chemical probes, and genome engineering. He received his PhD in 2011 from The Scripps Research Institute under the guidance of Prof. Benjamin F. Cravatt. From 2011–2015, he was a postdoctoral fellow in Prof. Todd Golub’s laboratory at The Broad Institute. Prof. Bachovchin joined TPCB and the Sloan Kettering faculty in September 2015. [Read more at: TPCB Faculty]
• Prof. Steven Gross of Weill Cornell Cornell is a leader in the use of mass spectrometry for the analysis of complex biological samples and has had a long-standing research focus on defining the mechanisms of cell signaling by nitric oxide. Most recently, he has established an untargeted metabolite profiling (aka metabolomics) platform to elucidate the broad consequences of genetic and chemical perturbations in vivo. Prof. Gross is an NIH MERIT Awardee and has been a member of the Weill Cornell faculty since 1988. [Read more at: TPCB Faculty]
• Prof. Xuejun Jiang of Memorial Sloan Kettering is a leader in studying the molecular basis of critical cellular processes involved in programmed cell death, autophagy, and regulation of tumor suppressors. His lab uses chemical genetic approaches to study new potential therapeutic targets and develops new small molecule inhibitors of those targets. Prof. Jiang has been a member of the Memorial Sloan Kettering faculty since 2003. [Read more at: TPCB Faculty]
• Prof. Shixin Liu of Rockefeller is a a biophysicist who studies the interaction, cooperation, and competition among molecular machines involved in gene regulation at the single-molecule level and genome wide. He completed his PhD in chemistry at Harvard in 2009, working with Prof. Xiaowei Zhuang, and has carried out his postdoctoral studies at UC Berkeley with Prof. Carlos Bustamante. Prof. Liu will join TPCB and establish the Laboratory of Nanoscale Biophysics and Biochemistry at Rockefeller in January 2016. [Read more at: TPCB Faculty]
TPCB Welcomes Incoming Class of 2015
TPCB welcomed eleven new students to the program: Ashley Chui (Cal State Fullerton), Hatice Didar Ciftci (Boğaziçi University), Emma Garst (Mount Holyoke College), Eun Bin Go (Harvey Mudd College), Darren Johnson (Townson University), Jonghan Lee (Hong Kong University), Rachel Leicher (Wesleyan University), Fangyu Liu (McGill University), Linamarie Miller (Williams College), Nikita Petukhov (Rice University), and Zheng Ser (Cornell University). Our new students represent diverse backgrounds and research interests in chemical biology and have started their first laboratory rotations.
May 2015
Olga Boudker Named HHMI Investigator
TPCB faculty member Prof. Olga Boudker of Weill Cornell Medical College has been selected as a Howard Hughes Medical Institute Investigator. She is one of 26 selected in the recent competition from nearly 900 applicants. Prof. Boudker’s laboratory uses structural, biophysical, and protein engineering techniques to understanding the mechanisms of membrane transporters. She joins TPCB’s other five HHMI investigators: Jue Chen, Jonathan Goldberg, Christopher Lima, Roderick MacKinnon, and Nikola Pavletich. [Read more at: Weill Cornell News | HHMI]
Adam Levinson Receives Honorable Mention for 2015 Rachele Prize
Congratulations to TPCB student Adam Levinson who received an Honorable Mention for the Julian R. Rachele research paper prize! Established in 1983, the Rachele Prize is the highest award given to students by the Weill Cornell Graduate School and is awarded to a PhD candidate for an original research paper on which the candidate is a major author. Adam was recognized for his sole-author paper describing the total synthesis of the alkaloid natural product aspeverin, published in Organic Letters. He is now working on the total chemical synthesis of the K-Ras protein. Adam completed his BS degree in Chemistry at Emory University and is carrying out his PhD studies with Prof. Samuel Danishefsky at Sloan Kettering. [Read the article in: Org. Lett.]
TPCB Congratulates Our 2015 Graduates
Congratulations to the TPCB’s 2015 graduates Gil Blum (Luo Lab, Sloan Kettering), Ian Bothwell (Luo Lab, Sloan Kettering), Roman Subbotin (Chait Lab, Rockefeller) and He Tian (Sakmar Lab, Rockefeller). Our graduates received their degrees at the Weill Cornell Graduate School of Medical Sciences graduation ceremony on May 28, 2015 at Carnegie Hall. Students in Sloan Kettering labs were also recognized at the Gerstner Sloan Kettering Commencement Ceremony on May 21, 2015. Students in Rockefeller labs will be recognized at The Rockefeller University Commencement Ceremony on June 11, 2015.
• Gil Blum, PhD – “Remodeling SETD8: A Holistic Approach for the Study of Protein Methyl Transferases (PMT’s)”, Postdoctoral Fellow, Prof. Minkui Luo, Sloan Kettering Institute
• Ian Bothwell, PhD – “Development and Application of Chemical Tools for the Study of SAM-Dependent Methyltranferases”, Postdoctoral Fellow, Prof. Wilfred van der Donk, University of Illinois at Urbana Champaign
• Roman Subbotin, PhD – “A Method for Determining Protein-Protein Interactions and Proximities in the Cellular Milieu”, Postdoctoral Fellow, Prof. Brian Chait, The Rockefeller University
• He Tian, PhD – “Development of Novel Chemical Biology Tools for Probing Structure-Function Relationship in G Protein Coupled Receptors”, Postdoctoral Fellow, Prof. Thomas Sakmar, The Rockefeller University
NYAS 2015 Chemical Biology Symposium Brings Together Labs from NYC and Beyond
TPCB partnered once again with the New York Academy of Sciences to present the annual NYAS Chemical Biology Symposium on May 18, 2015. The meeting is presented by the NYAS Chemical Biology Discussion Group, which brings together students, postdocs, and faculty from the NYC area and beyond to discuss the latest research in chemical biology. This year’s symposium featured keynote speaker Prof. Scott J. Miller of Yale University, who presented his lab’s latest work in catalyst development for the selective functionalization of complex molecules. TPCB students also presented their work in a vibrant poster session, alongside colleagues from Columbia, Yale, Princeton, U. Penn, NYU, Stony Brook, Hunter College, Mount Sinai, Albert Einstein College of Medicine, U. Delaware, Rutgers, and Seton Hall. [Read more at: NYAS]
April 2015
TPCB Founding Leader Joins Weill Cornell Medical College to Advance Cancer Research
Dr. Harold Varmus, co-founder of the TPCB program, joins Weill Cornell Medical College’s faculty as the Lewis Thomas University Professor of Medicine and will collaborate with investigators at the Sandra and Edward Meyer Cancer Center, led by Meyer Director and TPCB faculty member Prof. Lewis C. Cantley. Dr. Varmus was the President of the Memorial Sloan Kettering Cancer Center from 2000–2010 and was instrumental in the founding of the Tri-Institutional Research Program. He most recently Director of the National Cancer Institute until March 2015. Dr. Varmus was awarded the Nobel Prize in Medicine with J. Michael Bishop in 1989 for the discovery of the cellular origin of retroviral oncogenes. [Read more at: Weill Cornell News]
Stewart Shuman Elected Fellow of the American Academy of Arts and Sciences
TPCB faculty member Prof. Stewart Shuman at Memorial Sloan Kettering has been elected to the American Academy of Arts and Sciences. Prof. Shuman’s lab has led important research into the evolution and mechanisms of viral and microbial enzymes involved in the metabolism of nucleic acids, including processes such as mRNA synthesis and processing, and DNA repair and recombination. His wide-ranging discoveries have shed light on some of the most basic aspects of molecular biology, including how cells safeguard their genetic material and how genes are regulated. He joins five other TPCB faculty members as Fellows: C. David Allis (2001), Lewis Cantley (1999), Samuel Danishefsky (1984), Dinshaw Patel (2014), and Nikola Pavletich (2014). [Read more at: AAAS]
David Iaea Awarded NIH Fellowship
Congratulations to TPCB student David Iaea, who has been awarded a Ruth Kirschstein Predoctoral Fellowship from the National Institutes of Health! This fellowship will support David’s research on the structure and function of the sterol transport protein STARD4 with TPCB faculty Prof. Fred Maxfield at Weill Cornell. David has a Bachelor’s degree in Biochemistry from New York University and is interested in studying the mechanisms that mediate cholesterol distribution and homeostasis.
March 2015
Michaelyn Lux and Chaya Stern Awarded NSF Graduate Fellowships
Congratulations to TPCB students Michaelyn Lux and Chaya Stern who have been awarded a National Science Foundation (NSF) Graduate Research Fellowship. They join a select group of 2,000 awardees among 16,500 applicants nationwide. Michaelyn Lux is working with Prof. Derek Tan at Sloan Kettering on the synthesis of novel anticancer agents inspired by natural products. Michaelyn received her Bachelor’s degree in Chemistry from Michigan State University and is interested in studying organic chemistry and its applications to biological problems. Chaya Stern is working with Prof. John Chodera at Sloan Kettering in collaboration with Prof. Harel Weinstein and Prof. Scott Blanchard at Weill Cornell on combining single-molecule experiments with massively distributed molecular dynamics simulations to study G-protein coupled receptor dynamics. Chaya received her Bachelor’s degree in Chemistry from Brooklyn College and is interested in combining theory and experiments within a quantitative and testable framework to study biological macromolecules. [Read more at: NSF]
Lewis Cantley Wins 2015 Canada Gairdner International Award
TPCB faculty member Prof. Lewis Cantley of Weill Cornell Medical College has won the 2015 Canada Gairdner International Award from the Gairdner Foundation for his groundbreaking discovery of phosphoinositide 3-kinase (PI3K) and the signaling pathway that it controls. The Canada Gairdner International Awards are given annually to five biomedical scientists from around the world whose significant contributions to medicine have increased the understanding of human biology and disease. [Read more at: Weill Cornell News]
Minkui Luo Receives 2015 Eli Lilly Award in Biological Chemistry
TPCB faculty member Prof. Minkui Luo of Sloan Kettering has been awarded the 2015 Eli Lilly Award in Biological Chemistry from the American Chemical Society in recognition of his contributions to implement chemical tools to study biologically relevant enzymes. Prof. Luo’s focuses on functional annotation and pharmacological perturbation of protein methyltransferases (PMTs). The award recognizes his accomplishments in developing the novel Bioorthogonal Profiling of Protein Methylation (BPPM) technology to profile proteome-wide/genome-wide methylation events of designated PMTs; and leveraging unconventional strategies to access PMT inhibitors within the scope of PMT perturbation in general and disease therapy in particular. [Read more at: ACS]
February 2015
TPCB Faculty Collaboration Reveal New Insights into the Function of Transporters in the Brain
Healthy brain function depends upon communication between neurons carried out by neurotransmitters. After a neuron in the brain communicates with a neighboring neuron by releasing a burst of the neurotransmitter glutamate, those molecules must be cleared away rapidly to shut off the signal and ready cells for the next communication. Impaired glutamate transporter function is linked to neurodegeneration, brain damage following stroke and other disorders. TPCB faculty members Prof. Scott Blanchard and Prof. Olga Boudker and their labs at Weill Cornell have used a combination of single-molecule, X-ray crystallographic, and computational techniques to study glutamate transporters that shuttle this neurotransmitter across the cell membrane. Strikingly, they discovered that the glutamate transporter operates like an elevator, with a large portion of the protein gliding back and forth inside a membrane-embedded scaffold as it shuttles its cargo. This “elevator model” is entirely different from how other transporters were known to function. Their discoveries were made possible through collaborative efforts with Prof. Harel Weinstein at Weill Cornell and Prof. Jack Freed at Cornell University. [Read more at: Weill Cornell News| Read the article in: Nature]
January 2015
John Blenis, Olga Boudker, and Jue Chen Join the TPCB Faculty
TPCB recently welcomed three new faculty members to the program, bringing diverse research interests in chemical cell biology, small-molecule inhibitor discovery, and structure and function of membrane transporter proteins.
• Prof. John Blenis of Weill Cornell is a leader in the field of signal transduction and has helped to define major signaling pathways in cancer, including those involving Ras, PI3K, and mTOR. These studies have identified promising new therapeutic targets and the lab is now pursuing small-molecule screens to develop drug combinations that selectively kill cancer cells. Prior to joining Weill Cornell and TPCB, Prof. Blenis was on the faculty of Harvard Medical School for 25 years. [Read more at: TPCB Faculty]
• Prof. Olga Boudker of Weill Cornell has carried out seminal studies of the structure and function of glutamate transporters, which are responsible for the uptake of the neurotransmitter glutamate to enable proper synaptic function. These transporters use gradients of sodium and potassium ions to provide the necessary energy for glutamate transport. The lab uses X-ray crystallography, biochemistry, and biophysics to understand this coupled transport system. Prof. Boudker has been a member of the Weill Cornell faculty since 2005. [Read more at: TPCB Faculty]
• Prof. Jue Chen of Rockefeller is a structural biologist who is widely recognized for her studies of ABC transporters, which couple membrane transport of various molecules with ATP hydrolysis. Using X-ray crystallography, she has determined how this chemical energy is converted into mechanical work through a series of conformational changes in the transporter. Prof. Chen began her career at Purdue University in 2002 and was recruited to Rockefeller in 2014. She has also been an HHMI Investigator since 2008. [Read more at: TPCB Faculty]
November 2014
David Allis Wins 2015 Breakthrough Prize for Discovery of Histone Modifications
TPCB faculty member Prof. C. David Allis of Rockefeller has been awarded one of six 2015 Breakthrough Prizes in Life Sciences for his seminal research in epigenetics. Prof. Allis was recognized for his discovery of covalent chemical modifications of histone proteins that package DNA, and the critical roles of these modifications in regulating gene expression. This fundamentally new mechanism for gene regulation has major implications in a variety of human diseases including cancer and opens the door to the development of new therapeutic approaches. Prof. Allis joins Prof. Lewis Cantley of Weill Cornell as the second TPCB faculty member to be honored with this award. In total, six faculty on the Tri-Institutional campuses have received Breakthrough Prizes, which were established in 2013 by technology entrepreneurs including Google’s Sergey Brin, Facebook’s Mark Zuckerberg, and Alibaba’s Jack Ma and are accompanied by cash awards of $3 million. [Read more at: Rockefeller News]
Lewis Cantley Elected to Institute of Medicine
TPCB faculty member Prof. Lewis Cantley at Weill Cornell has been elected to the Institute of Medicine of the National Academies. Membership in the IOM is one of the highest honors bestowed in the fields of health and medicine. Prof. Cantley is a preeminent chemical biologist and cancer researcher who discovered the phosphoinositide 3-kinase (PI3K) signaling pathway. PI3K is the most commonly mutated gene across various cancers and this discovery has led to revolutionary treatments for cancer, diabetes, and autoimmune diseases. His research focuses on understanding the biochemical pathways by which oncogenes mediate changes in cell metabolism, cell growth, and cell transformation. Prof. Cantley joined Weill Cornell and the TPCB faculty in 2012 from Harvard Medical School and is also Director of the Meyer Cancer Center. [Read more at: Weill Cornell News]
October 2014
Marraffini Lab Develops Programmable Antibiotics to Target Bad Bugs
Most antibiotics used clinically are blunt instruments, which kill not only pathogenic bacteria but also commensal bacteria, often with serious side effects. To address this problem, TPCB faculty member Prof. Luciano Marraffini and his lab at Rockefeller have now developed a smarter, ‘programmable’ antibiotic strategy that selectively targets pathogenic bacteria, particularly those carrying genes for antibiotic resistance. In the approach, they engineer the CRISPR-Cas9 system, which bacteria use to defend themselves against viruses, to target specific DNA sequences for cleavage. By targeting bacterial gene sequences known to encode for antibiotic resistance, this approach could address the most life-threatening infections while simultaneously sparing commensal bacteria, thereby avoiding secondary infections such as those caused by Clostridum difficile. The work demonstrated that the antibiotic system could be used to kill resistant Staphylococcus aureus in a mouse skin infection model and was carried out in collaboration with the lab of Rockefeller colleague, Prof. Vincent Fischetti. [Read more at: Rockefeller News | Read the article in: Nat. Biotechnol.]
August 2014
TPCB Co-Sponsors New York Academy of Sciences Symposium on Click Chemistry
TPCB was proud to co-sponsor the New York Academy of Sciences event, Click Chemistry in Biology and Medicine Symposium: New Developments and Strategies. The symposium was held on September 15, 2014 and explored recent developments in click chemistry, including the discovery of new click reactions. The program included keynote talks by 2001 Nobel Laureate Prof. K. Barry Sharpless and Prof. James Paulson, both of The Scripps Research Institute. Other speakers included Prof. Yimon Aye (Cornell University), Prof. David A. Spiegel (Yale University) and Prof. Peng Wu (Albert Einstein College of Medicine). [Read more at: NYAS]
July 2014
TPCB Symposium Showcases Forefront Research in Chemical Biology
TPCB hosted its 10th Annual Chemical Biology Symposium on July 18, 2014 at The Rockefeller University with over 100 chemical biology researchers in attendance. The program featured four distinguished keynote speakers: Prof. Raymond Deshaies from the California Institute of Technology, Prof. Kai Johnsson from École Polytechnique Fédérale de Lausanne, Prof. Dorothee Kern from Brandeis University, and TPCB faculty member Prof. Sebastian Klinge from Rockefeller University. TPCB students Ian Bothwell (Luo Lab, Sloan Kettering), Rashad Karimov (Gin/Tan Lab, Sloan Kettering), and Roman Subbotin (Chait Lab, Rockefeller) also presented seminars on their graduate research. Numerous scientists from the broader Tri-Institutional chemical biology community also shared their work at a poster session, with Best Poster Awards going to Gil Blum (Luo Lab, Sloan Kettering), David Iaea (Maxfield Lab, Weill Cornell), Alexis Jaramillo (Nimigean Lab, Weill Cornell), and Dr. Nathan Westcott (Hang Lab, Rockefeller).
June 2014
Kapoor Lab Develops New Approach to Identify Biological Targets of Small Molecules
Identifying the biological targets of bioactive small molecules is a major challenge in chemical biology and a classical bottleneck in the field. TPCB Faculty Member Prof. Tarun Kapoor and his lab at Rockefeller University have now developed a powerful new approach that enables rapid identification and validation of these targets, using state-of-the-art genome sequencing and editing technologies. The work was carried out through a multidisciplinary collaboration with computational biologist Prof. Olivier Elemento at Weill Cornell Medical College. Their approach involves initial identification of cells that are resistant to the activity of the small molecule, followed by high-throughput transcriptome sequencing to identify the genetic mutations in the cells that confer this resistance. These candidate targets are then validated by using a genome editing technology called the CRISPR-Cas9 system to insert the specific mutated gene into a new cell line and confirm resistance to the small molecule. The method can be used to identify both direct physiological targets of the molecules as well as epigenetic and other indirect resistance mechanisms. The new technology was demonstrated using two anticancer drugs and is called ‘DrugTargetSeqR’. [Read the article in: Nat. Chem. Biol.]
Pavletich and Patel Elected Fellows of the American Academy of Arts and Sciences
TPCB faculty members Prof. Nikola Pavletich and Prof. Dinshaw Patel at Memorial Sloan Kettering have been elected to the American Academy of Arts and Sciences. Prof. Pavletich’s research group is interested in the structural biology of pathways that are altered in cancer, with particular emphasis on pathways that control the growth and proliferation of the cell. Prof. Patel’s lab applies crystallographic and solution NMR techniques to investigate macromolecular-mediated recognition, regulation, and catalysis. They join two other TPCB faculty members as Fellows, Prof. Samuel Danishefsky (1984) and Prof. C. David Allis (2001). [Read more at: AAAS]
NYAS Chemical Biology Symposium Features Local Students and Postdocs
TPCB partnered with the New York Academy of Sciences to present the annual year-end NYAS Chemical Biology Symposium on June 3, 2014. The meeting is a highlight of the NYAS Chemical Biology Discussion Group series, which aims to enhance interactions among local-area laboratories working in chemical biology and to feature forefront research in chemical biology to the wider community. The series traditionally covers a range of current topics in chemical biology, including chemical probe development, organic synthesis, biosynthesis, protein engineering, nanotechnology, and drug discovery. This year’s symposium featured keynote speaker Prof. Brent Stockwell of Columbia University, as well as student talks and posters from participating tri-state area institutions, including several TPCB labs. [Read more at: NYAS]
May 2014
TPCB Congratulates Our 2014 Graduates
Congratulations to the TPCB’s 2014 graduates Anupam Chakravarty (Shuman Lab, Sloan Kettering), Ushati Das Chakravarty (Shuman Lab, Sloan Kettering), Rashad Karimov (Gin/Tan Lab, Sloan Kettering), and Vidhya Rangaraju (Ryan Lab, Weill Cornell)! The graduates received their degrees at the Weill Cornell Graduate School of Medical Sciences graduation ceremony on May 29, 2014 at Carnegie Hall. Students in Sloan Kettering labs were also recognized at the Sloan Kettering Institute Commencement Ceremony on May 14, 2014.
• Anupam Chakravarty, PhD – “Mechanisms of RNA Damage and Repair”, Postdoctoral Fellow, Prof. Daniel Jarosz, Stanford School of Medicine
• Ushati Das Chakravarty, PhD – “End-healing Mechanisms in DNA and RNA Repair”, Postdoctoral Fellow, Prof. Harry Noller, University of California Santa Cruz
• Rashad Karimov, PhD – “Synthetic Studies on Immunoadjuvant Jujuboside A. Total Synthesis of 23-epi-Jujubogenin”, Postdoctoral Fellow, Prof. John F. Hartwig, University of California, Berkeley
• Vidhya Rangaraju, PhD – “Developing a Novel Quantitative ATP Reporter and Dissecting the Role of ATP in Synaptic Function”, Postdoctoral Fellow, Prof. Erin Schuman, Max Planck Institute for Brain Research
Sebastian Klinge awarded Alfred P. Sloan Research Fellowship
TPCB faculty member Prof. Sebastian Klinge of Rockefeller University was one of 126 scholars nationwide selected to receive an Alfred P. Sloan Research Fellowship. The fellowship supports early-stage scientists pursuing original research. Prof. Klinge will receive $50,000 over two years to further his work on the structure and function of ribosomes, the cell’s protein factories. Three TPCB faculty have been awarded Sloan fellowships previously: Prof. Vanessa Ruta (2013), Prof. Derek Tan (2007), and Prof. Timothy Ryan (1999). [Read more at: Rockefeller News]
April 2014
Adam Trotta Awarded NSF Graduate Fellowship
Congratulations to TPCB student Adam Trotta, who has been awarded a National Science Foundation (NSF) Graduate Research Fellowship to support his research on synthesis of antibacterial alkaloid natural products with Prof. Samuel Danishefsky at Sloan Kettering! Adam has a Bachelor’s degree in Chemistry from Tufts University and is interested in studying efficient total synthesis of bioactive natural products. He is one of a select group of 2,000 students to receive this honor out of 14,000 applicants nationwide. [Read more at: NSF]
March 2014
Tiny Solutions for Big Problems: A Visit to the Lab of Daniel Heller
Chemist and engineer Prof. Daniel Heller, a TPCB faculty member, makes nanoscale materials that are specially designed to enable biological research and solve clinical problems. Using their diverse sets of expertise, members of his lab at Memorial Sloan Kettering work closely together to speed developments in cancer research, diagnosis, and treatment. Team members in the Heller lab are developing carbon nanotube–based sensors to detect early-stage cancers, as well as nanoparticles to target drugs that treat metastatic tumors. [Read more at: MSK News | View video]
February 2014
Jaffrey Lab Uncovers Mechanism Involved in Fragile X Syndrome
TPCB faculty member Prof. Samie Jaffrey and his lab at Weill Cornell have uncovered the mechanism by which RNA can direct gene silencing involved in fragile X syndrome, a genetic condition that causes intellectual disability and autism. In their paper in Science, they report that an untranslated region of the messenger RNA forms an RNA•DNA duplex with its complementary DNA strand of the fragile X syndrome gene, shutting down production of a protein needed for communication between brain cells. The researchers also discovered that a small molecule that binds this untranslated region of the mRNA can block this gene silencing mechanism in human embryonic stem cells that carry the allele for fragile X syndrome. This suggests that similar therapeutic strategies may be possible for 20 other diseases caused by related gene silencing mechanisms, ranging from mental retardation to multisystem failure. [Read more at: Weill Cornell News | Read the article in: Science]
Tim Ryan and Vidhya Rangaraju Discover New Paradigm for ATP Production in Neurons
TPCB student Vidhya Rangaraju and TPCB faculty member Prof. Timothy Ryan at Weill Cornell have used a novel molecular imaging technology to challenge the conventional dogma on energy production in neurons. Scientists have long thought that neurons produce a steady stream of ATP to enable neurotransmission. However, in their latest paper in Cell, the Ryan lab used a quantitative luciferase reporter to monitor presynaptic ATP levels and discovered that ATP is produced on-demand, rather than continuously. This suggests that brain disorders may be caused by mutations or malfunctions in this on-demand circuitry. Further research will be required to see if such ATP deficits occur in diseased states but this research suggests that new therapeutic approaches might be considered to treat neurodegenerative disorders. [Read more at: Weill Cornell News | Read the article in: Cell]
January 2014
Davis Allis Receives 2014 Japan Prize in Life Sciences
TPCB faculty member Prof. C. David Allis of Rockefeller University has been awarded the 2014 Japan Prize in Life Sciences! This prestigious international award is presented to individuals whose original and outstanding achievements are scientifically impressive, and have also served to promote peace and prosperity for all mankind. Prof. Allis is a pioneer in the field of epigenetics and is being recognized for his discovery of the chemical modifications on DNA-packaging histone proteins that serve as fundamental regulators of gene expression. Each Japan Prize laureate receives a cash prize of 50 million yen. Congratulations to Prof. Allis and his laboratory! [Read more at: Rockefeller News | Japan Prize Foundation]
Danishefsky Lab Completes Total Synthesis of the Glycoprotein Erythropoietin
TPCB faculty member Prof. Samuel Danishefsky of Sloan–Kettering and his team have reported a landmark achievement in Science, bridging the gap between biologics and small-molecule drugs by accomplishing the total chemical synthesis of the glycoprotein erythropoietin (EPO) in a single, pure form. EPO is the hormone that controls the production of red blood cells by a process called erythropoiesis. The ability to generate pure EPO will now enable scientists to make numerous forms of the molecule and allow them to study whether certain variants are better than others at inducing the production of red blood cells, as well as participating in other biological processes. The achievement required major advances in the logic of building oligosaccharides and new strategies for assembling polypetides and proteins in a chemical laboratory rather than in a biological cell. The work had been highlighted in both Science and Nature. [Read the article in: Science | Read highlights in: Science | Nature]
December 2013
Vanessa Ruta Receives 2013 NIH Directors’ New Innovator Award
TPCB faculty member Prof. Vanessa Ruta of Rockefeller University was one of 41 investigators nationwide selected to receive an NIH Directors’ New Innovator Award. The grant supports early-stage investigators pursuing bold and highly innovative research projects. Dr. Ruta is working on a novel neural tracing technique to map the associative olfactory circuits in the fly brain and to discern how odor associations are encoded by synaptic connections and neural ensembles. Her goal is to use optical imaging, labeling, and electrophysiology to directly visualize how individual synaptic connections between neurons are modified as a consequence of learning and experience. The grant provides $2.45 million in funding over a five-year period. Congratulations to Prof. Ruta and her laboratory! [Read more at: NIH]
Michael Foley to Lead Tri-Institutional Therapeutics Discovery Institute
Preeminent chemist and entrepreneur, Michael A. Foley, PhD, has been named Director of the new Tri-Institutional Therapeutics Discovery Institute. This innovative partnership of Weill Cornell, Rockefeller, Sloan-Kettering, and Takeda Pharmaceutical Company is aimed at advancing early-stage academic drug discovery and will be housed in the top floor of the new Belfer Research Building at Weill Cornell. Dr. Foley was most recently the Director of the Chemical Biology Platform at the Broad Institute of MIT and Harvard. He is the scientific co-founder of four biotechnology companies, a veteran of the pharmaceutical industry, and has placed 12 drugs into clinical development. [Tri-I TDI | Read more at: Weill Cornell Bews | Rockefeller News | MSK News]
Aconitine Alkaloid Natural Product Total Synthesis Highlighted
Congratulations to TPCB alumnus Dr. Yuan Shi, whose paper in the Journal of the American Chemical Society describing the first total synthesis of the norditerpenoid alkaloid natural product neofinaconitine has been highlighted in Angewandte Chemie as well as in Synfacts. The work was carried out with Dr. Jeremy Wilmot and Dr. Lars Nordstrøm in the laboratory of the late Prof. David Y. Gin, a TPCB faculty member. [Read the article in: J. Am. Chem. Soc. | Read highlights in: Angew. Chem. Intl. Ed. | Synfacts | Org. Chem. Highlights]
Howard Hang and Minkui Luo Speak at Bioorthogonal Chemistry Symposium
TPCB faculty members Prof. Howard Hang and Prof. Minkui Luo, along with Prof. Joe Fox (University of Delaware) presented their research at the New York Academy of Sciences symposium on “Bioorthogonal Chemistry in Biology and Medicine” on December 11, 2013. The symposium featured a stimulating afternoon of lectures and posters with numerous labs from the NYC area in attendance. [Read more at: NYAS]
November 2013
Joan Massagué Named Director of Sloan-Kettering Institute
Renowned cancer biologist Joan Massagué, PhD has been named Director of the Sloan-Kettering Institute, succeeding Dr. Thomas J. Kelly. With a research career spanning more than 30 years, Dr. Massagué has been lauded for the originality and importance of his work elucidating the mechanism of action for transforming growth factor-beta (TGF-β) signaling. He joined MSKCC in 1989 as Chair of the Cell Biology Program and was named the inaugural Chair of the Cancer Biology & Genetics Program in 2003. He is a member of the National Academy of Sciences and the Institute of Medicine, and an Investigator at the Howard Hughes Medical Institute. [Read more at: MSK News]
Vidhya Rangaraju Completes Her PhD Thesis on a Novel Optical ATP Reporter
Congratulations to TPCB student, Vidhya Rangaraju, who successfully defended her thesis “Developing a Novel Quantitative ATP Reporter and Dissecting the Role of ATP in Synaptic Function” on November 14, 2013! Vidhya carried out her research in the lab of TPCB faculty member Prof. Timothy Ryan at Weill Cornell, where she developed a novel, genetically-encoded, optical ATP reporter for quantitative dynamic measurements of presynaptic ATP concentrations. Using this reporter, she found that electrical activity causes significant demands on presynaptic ATP levels that are met by activity-driven ATP synthesis. The synaptic vesicle cycle serves as the primary source of this ATP demand, resulting in severe impairment of synapse function when local ATP synthesis is compromised (Rangaraju, V.; Calloway, N.; Ryan, T. A. “Activity-driven local ATP synthesis is required for synaptic function.” Cell 2013, in press). She will now be going onto a postdoctoral fellowship with Prof. Erin Schuman, Director of the Max Planck Institute for Brain Research in Frankfurt, Germany. Herzlichen Glückwunsch, Vidhya!
October 2013
Tri-Institutional Therapeutics Discovery Institute to Accelerate Drug Development
Memorial Sloan-Kettering Cancer Center, The Rockefeller University and Weill Cornell Medical College have formed the pioneering Tri-Institutional Therapeutics Discovery Institute (Tri-I TDI) in partnership with Takeda Pharmaceutical Company. This new, groundbreaking institute is designed to expedite the translation of early-stage drug discoveries into innovative treatments and therapies for patients. The Tri-I TDI has been founded thanks to a generous $15 million gift by Lewis and Ali Sanders and a $5 million gift from Howard and Abby Milstein. [Read more at: MSK News | View video]
September 2013
TPCB Symposium Features Leading Chemical Biologists
TPCB hosted its 9th Annual Symposium on September 30, 2013 at Sloan-Kettering. The Symposium program featured four distinguished keynote speakers: Prof. Andrew Phillips (Yale University), Prof. Hening Lin (Cornell University), Prof. Chuan He (University of Chicago) and TPCB faculty member Prof. Samie Jaffrey (Weill Cornell Medical College). Current TPCB students Ushati Das and Qinsi Zheng also presented seminars on their research and a lunchtime poster session included numerous participants from the broader Tri-Institutional chemical biology community.
Ushati Das Awarded Hutchinson Fellowship
Congratulations to TPCB student Ushati Das, who received the Dorris J. Hutchison Graduate Fellowship to support her research on the structures and mechanisms of RNA repair enzymes with Prof. Stewart Shuman at Sloan-Kettering! [Read more at: MSK News]
Gary Koretzky Named Dean of Weill Cornell Graduate School of Medical Sciences
One of the world’s leading immunologists, Gary Koretzky, MD, PhD, has been named Dean of the Weill Cornell Graduate School of Medical Sciences and Senior Associate Dean for Research at Weill Cornell Medical College, effective October 1, 2013. He succeeds Dr. David Hajjar and joins Dean Sidney Strickland of Rockefeller University and Dr. Thomas Kelly of Memorial Sloan-Kettering Cancer Center on the Executive Committee of TPCB. Dr. Koretzky has been the Francis C. Wood Professor of Medicine at the University of Pennsylvania School of Medicine and studies the biochemistry of T lymphocyte development. He is a member of the Institute of Medicine and a Fellow of the American Academy of Arts and Sciences. [Read more at: Weill Cornell News]
July 2013
TPCB Welcomes Incoming Class of 2013
TPCB welcomed six new students to the program: Shi Chen (Tsinghua University), Malik Chaker-Margot (University of Montreal), Zachary Hann (University of California, San Diego), Cristina Santarossa (Smith College), Chaya Stern (Brooklyn College), and John Zinder (Cornell University and University of Colorado). These students represent diverse research interests in chemical biology and have started their first laboratory rotations. [Read more at: TPCB Students]
May 2013
Christopher Lima Selected as HHMI Investigator
TPCB faculty member Prof. Christopher D. Lima of Sloan-Kettering’s Structural Biology Program has been selected as a Howard Hughes Medical Institute Investigator. He is one of 27 selected in the recent competition from more than 1,200 applicants. Dr. Lima’s laboratory uses structural, biochemical, and functional analyses of macromolecules to understand RNA metabolism and its role in cell growth and disease. Dr. Lima joins TPCB’s three other HHMI investigators: Jonathan Goldberg, Roderick MacKinnon, and Nikola Pavletich, along with HHMI Early Career Scientist Sean Brady. [Read more at: MSK News]


